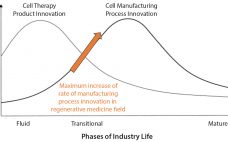
Most commercial biopharmaceuticals originated from academic research laboratories and start-up development laboratories. Despite such products having differences in modalities and targeted disease indications, and whether their target patient populations are relatively small or approaching blockbuster status, at a key point in development, biopharmaceutical production must scale up from laboratory to commercial production. That movement from research to development and then to manufacturing forces attention on economics and speed to market, and it drives innovative approaches to producing biopharmaceutical cell compositions…
Upstream Processing
Cell Culture Scale-Up in Stirred-Tank Single-Use Bioreactors
Bioprocess development usually is carried out in systems with small working volumes. This helps save time and resources because, at small scale, several experiments can be conducted in parallel. Costs for media are kept low, and relatively little laboratory space is required to operate small-scale bioreactors. But over the course of development, biopharmaceutical companies need more material for characterization, trial runs, and finally for commercialization. They transition to bench scale and then up to pilot or production scale with the…
Improving Bioreactor Performance Measuring Dissolved Oxygen to Determine kLa
In recent years the biopharmaceutical industry has significantly increased the demands it makes on bioreactor systems. Efficient and reproducible production of active pharmaceuticals of high quality and in large quantities is of highest priority. However, bioprocessing is a complex topic. Numerous factors affect growth of cells in culture but are difficult to determine and interpret reliably. One of the most relevant performance parameters is the volumetric mass transfer coefficient (kLa). It describes the efficiency of gas transfer (e.g., oxygen) from…
Cell Culture Media: An Active Pharmaceutical Ingredient or Ancillary Material?
Cell-based therapies are used to treat diseases that require the replacement of diseased, dysfunctional, and injured cells (1). To produce these therapies, a wide range of reagents and materials such as antibodies, growth factors, and enzymes are used in their manufacturing processes. Such necessary materials are administered through a cell culture medium. Active pharmaceutical ingredients (APIs) are the main ingredients that make products therapeutic. Ancillary materials (AMs) and raw materials (RMs) are essential components used during production but are not…
Single-Use Bioreactors: Performance and Usability Considerations, Part 2
As the biopharmaceutical industry continues toward streamlined bioprocessing and intensified cell-culture biology, selection criteria of single-use bioreactors (S.U.B.s) and other bioprocessing technologies will become increasingly rigorous, emphasizing the importance of considering every aspect of technologies under evaluation. In part 1, we discussed performance for process control, including the maintenance of critical process parameters (CPPs), and highlighted bioreactor performance (e.g., mass transfer, power per volume, and temperature control) as a critical consideration during the selection of S.U.B.s (1). Part 2 focuses…
Assurance of Clonality: Next-Generation Single-Cell Dispensing in Cell Line Development and Single-Cell Genomics
At the Cell Line Development and Engineering (CLD&E) conference(23–25 April 2018, Amsterdam), Jonas Schöndube, CEO of cytena GmbH, gave a presentation highlighting some of the company’s recent developments in single-cell dispensing for documented clonal cell lines. About cytena cytena is a young company dedicated to the development and manufacture of tools for the biopharmaceutical industry. In 2015, cytena launched the single-cell printer™ (scp™), which enables fully automated isolation of single cells into 96- and 384-well plates. The patented technology uses…
Oxygen Mass Transfer Correlation for a Rocking-Motion Bioreactor System
Disposable bioreactor systems are technologies commonly used in bioprocessing. They provide cost-effective contamination control and allow more flexible facility layouts than do stainless steel alternatives. One of the most popular types of single-use bioreactors uses a rocking platform in place of a traditional shaft and agitator assembly to aerate and mix cell culture material within a presterilized, single-use plastic bag (1). The system studied here is the ReadyToProcess WAVE 25 bioreactor (GE Healthcare Life Sciences). In contrast to conventional stirred…
Single-Use Bioreactors: Performance and Usability Considerations Part 1: Performance for Process Control
There is ever increasing pressure for the biopharmaceutical industry to drive toward higher efficiency and lower costs. Compared to the past, target markets for many drugs typically are becoming smaller, and so-called blockbuster drugs are becoming more the exception than the rule. Regulatory agencies have continued to increase the pressure on drug makers to meet increasing quality standards and accept higher levels of responsibility. Furthermore, customer pricing, healthcare markets, and recent biopharmaceutical pricing scandals all add incentives toward more efficient…
eBook: Raw Materials Quality, Processing, and Storage — A Manufacturing Case Study
Raw material storage, handling, and processing are essential to ensure high product quality and consistent process performance. Slight variabilities in raw materials (either inherent in the material or through processing) can compromise yield and even result in batch loss. On Tuesday 26 September 2017 speakers at the BioProcess International Conference (part of Biotech Week Boston) addressed raw material variability and control strategies in biomanufacturing. They discussed the industry’s initiative for raw material risk assessments and strategies to control variability by…
eBook: The Commercial Expression Systems Market — What Has Changed in the Past Decade
A decade ago, BioPlan Associates prepared the findings of its 2008 directory of expression system technologies that were being promoted or considered likely to be suitable for commercial licensing for biopharmaceutical manufacturing (1). Due in part to the relatively slow advances in this critical area of bioprocessing, this study remains perhaps the only directory of biopharmaceutical-relevant expression systems available for licensing. Here I discuss aspects of related bioprocessing technologies that have and have not changed in the past decade. Expression…