Scalability remains a critically important topic for biopharmaceutical companies. For conventional protein products, the strategy once was straightforward: Drug makers would scale up, beginning with cultures in flasks and roller bottles to grow enough cells to inoculate laboratory-scale (often glass) bioreactors, then again to pilot- and commercial-scale, stainless-steel, stirred-tank bioreactors. At their highest volumes, such reactors can handle tens of thousands of liters of cells and growth media. If a drug developer did not have the requisite equipment to scale…
Downstream Development
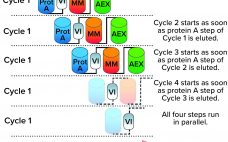
Reducing Downstream Scale-Up Needs: Advances Toward Continuous Downstream Processing
The biopharmaceutical industry generally acknowledges that upstream and downstream aspects of drug-substance manufacturing are experiencing a capacity mismatch. Today, many recombinant proteins can be produced at expression titers of 3 g/L, with some yields exceeding 10 g/L. Such titers represent 100-fold increases in production capability compared with values from twenty years ago (1, 2). Increases in cell-culture density and improvements to perfusion-mode bioreactor systems hold promise for increasing yields further still. Such developments, combined with the broad availability of concentrated…
Mind the Gap: Managing Relationships Between Upstream and Downstream Intensification
Process intensification (PI) describes an integrated framework of strategies to maximize the output of a unit operation, a process, or an entire facility. By implementing PI strategies, biomanufacturers can accomplish their productivity goals by increasing production speeds and titers, reducing facility footprints, and cutting costs. Overall, such changes improve production efficiency and flexibility. Collectively, the biotherapeutic industry has made multiple advancements in intensifying upstream processing. PI strategies include using high-density cell banks, implementing seed-train intensification (n – 1 perfusion), and…
Ask the Expert: HCP Analysis By Orthogonal Methods in Vaccine and Gene Therapy Development
Regulators require testing of drug products for process-related impurities throughout development to monitor product safety, purity, and efficacy. Low levels of most impurities can be inconsequential, but patient safety demands that host-cell proteins (HCPs) be eliminated or reduced to the lowest levels practical. Enzyme-linked immunosorbent assays (ELISAs) represent a key tool in that endeavor. Antibody-coverage analysis is one part of assessing a platform kit or custom HCP ELISA. In a 15 June 2021 webinar, Jared Isaac (senior scientist at Cygnus…
Accelerating the Development and Manufacture of Therapeutics Using the Octet Platform
The high costs of therapeutic discovery, development, and manufacture require improved process efficiencies and economics. Analytical tools that eliminate the need for reagent labeling and enable real-time data visualization save development time and improve efficiencies during process development. The Octet biolayer interferometry (BLI) platform and assays can be used throughout process development and manufacturing, including cell-line development, clone selection, and dynamic binding capacity (DBC) determination for affinity purification columns. The ability of the Octet BLI platform to monitor binding interactions…
Your Partner for Host Cell Protein Analytics for Over 25 Years
Cygnus Technologies, part of Maravai LifeSciences, is the biopharmaceutical industry’s partner in host cell protein (HCP) and other process-related impurity detection and analytics. In addition, Cygnus now provides innovative viral clearance solutions. Cygnus helps companies developing therapeutic proteins, vaccines, antibodies, plasma derivatives, and gene therapies ensure the safety of their biotherapeutics before human trials, regulatory approval, and commercial release. Cygnus provides analytical tools and solutions for improving bioprocess development for faster regulatory approval and better clinical outcomes. Cygnus is an…
Ask the Expert: Considerations for Mass Spectrometric HCP Analysis During Process Development
Host-cell proteins (HCPs) must be assessed early in biopharmaceutical product development to establish the efficiency of a purification process. Liquid chromatography coupled with mass spectrometry (LC-MS) is a strong orthogonal method for HCP identification and quantitation. During an 18 March 2021 “Ask the Expert” presentation, Christina Morris (senior scientist at BioPharmaSpec) explained how MS analyses can enhance process development (PD), then presented considerations for generating spectral libraries, selecting samples for quantitation, and reviewing data. Morris’s Presentation Spectral Libraries: A typical…
Endotoxin Questions and Answers
Endotoxin — also known as lipopolysaccharide (LPS) — is a large molecule made up of a lipid and a polysaccharide portion found in the outer membrane of each Gram-negative bacterial cell. Examination of endotoxins is necessary for microbial and animal cell-culture operations that produce active pharmaceutical ingredients (APIs) and vaccines. If a cell culture is contaminated with endotoxins, it could affect cell growth and function (thus potentially affecting product quality). Biopharmaceutical companies need to understand the endotoxin levels within their…
eBook: Challenges in Industrial Process Development of Exosome-Based Therapies: Characterizing and Managing Diversity
The traditional classification of extracellular vesicles (EVs) includes three types: exosomes, microvesicles, and apoptotic vesicles. Each type arises from a distinct origin and exhibits distinct characteristics. The problem is that their size ranges overlap and that the major surface proteins presented by exosomes also are present on the surfaces of microvesicles and apoptotic bodies. This makes it a challenge for process developers to identify the vesicle fraction that best serves a particular exosome therapy. Anion-exchange chromatography (AEC) can fractionate EVs…
The Downstream Perspective: Putting Product Knowledge to Work Using Technological Innovations
After over a quarter century in the industry — including downstream processing (DSP) and manufacturing directorships at Boehringer Ingelheim and leadership roles in technology development, quality, and manufacturing at Novasep — European consultant Margit Holzer is a recognized expert in downstream processing of biopharmaceutical products. Holding a doctorate in biotechnology from the University of Natural Resources and Applied Life Sciences in Austria, Holzer is familiar to BPI readers as both an author and conference participant (1, 2). And in May…