Ever since the first biopharmaceutical product (biologic) was approved in the 1980s, companies have developed protocols and tests to ensure that such products are safe and effective. Biologics are very different from traditional small-molecule drugs, with unique risks inherent to their manufacturing processes. Biopharmaceutical formulations often present as complex mixtures that can be sensitive to heat, light, and many other factors, all of which must be monitored and assessed. However, until recently, developers worked mostly independently, with only their own…
Leachables/Extractables/Particulates
Integrity of Single-Use Systems: Practical Applications and Deployment
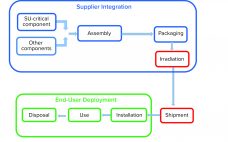
Single-use (SU) technology plays an important role in modern vaccine and biologics manufacturing. System integrity, managed by critical process controls, ensures sterility and is a prerequisite for successful leak-free processing. Nonintegral systems cause loss of product, quality, and time; increase costs through investigations; and lead to potential safety problems. The BioProcess Systems Alliance (BPSA) issued a white paper in 2017, Design, Control, and Monitoring of Single-Use Systems for Integrity Assurance (1), that describes in detail the strategies for design and…
The Proof Is in the Data: Extractables and Leachables
Extractables and leachables (E&Ls) must be addressed in material and process validation programs. Extractables are compounds that can be extracted from a material in the presence of solvents with varying polarity under extreme conditions. Materials manufacturers should make extractables guides available to end users. Leachables are compounds that migrate from a material in the presence of an actual formulation under normal process operating conditions. Extractables information can be helpful as a basis for evaluation of potential process-equipment–related leachables (PERLs)testing. However,…
Embedded Particles in Single-Use Bags: Risk to Bag Integrity and Drug Product Purity, or Only a Cosmetic Defect?
When using single-use systems (SUS) to process biopharmaceuticals, preventing drug product contamination from extractables and leachables (E&Ls) and embedded particulate matter (gel particles) in the polymer films used to make bioprocess bags is critical. Using a pressure burst test to assess film integrity, Sartorius Stedim Biotech’s Klaus Wormuth and colleagues compared Flexboy and Flexsafe samples with gel-particle-free materials to assess their potential for contamination. The results showed that only large (2–4 mm2) gel particles affected the burst test results, concluding…
Integrity Redefined: Consistent Robustness and Integrity Testing Lead to Enhanced Process Integrity and Patient Safety
With the increasing adoption of single-use systems (SUS) in critical stages of biopharmaceutical manufacturing, any lack of system integrity can significantly affect drug product quality and patient safety, as well as incur additional costs due to product loss and disrupted production cycle. This article from Sartorius Stedim Biotech, describes how determining the correlation between liquid leakage and microbial ingress can be used to define MALLs (Maximum Allowable Leakage Limits) of SUS for different process steps. The article also details the…
Visible Particulate Matter in Single-Use Bags: From Measurement to Prevention
Parenteral pharmaceuticals must be “essentially free” from visible particulate matter (1). In the production of biopharmaceuticals with single-use systems (SUS), biocompatibility requires controlling interactions between drug substances/products and SUS surfaces to ensure drug product quality and patient safety with regard to extractables/leachables and particulate matter. Any particulate matter stuck to fluid-contacting surfaces of process components could wash off and contaminate process fluids. Depending on system configuration, a final drug product could be at risk for particulate matter from SUS. Risk…
The Changing Landscape of Single Use in Bioprocessing: An Interview with Stefan Schlack and Jean-Marc Cappia
Single use in bioprocessing has changed significantly in recent years. To find out how a leading supplier to the biopharmaceutical industry is redefining its technology to align with new market challenges, science writer, Sue Pearson, had the opportunity to interview Stefan Schlack, Head of Bioprocess Marketing, and Jean-Marc Cappia, Head of Segment Marketing Vaccines, both at Sartorius Stedim Biotech (SSB) in Goettingen, Germany. Redefining Single Use Pearson: Sartorius Stedim Biotech is a recognized leader in single-use technology, and you’ve recently…
Development and Biomanufacturing Strategies for Next-Generation Antibody-Drug Conjugates
The development and manufacture of antibody-drug conjugates (ADCs) requires a series of complex steps. ADC manufacturers must comply with guidelines for both the small-molecule linker-drug and the monoclonal antibody (MAb). The authors describe their company’s development of its lead ADC product. They review process decisions, including the issues that factored into their selection of single-use systems, manufacturing challenges (differences between ADCs and MAbs), and testing methodologies for extractables and leachables. Synthon began as a small-molecule generics company in 1991. During…
Development of a Standardized Extractables Approach for Single-Use Components: General Considerations and Practical Aspects — A Manufacturer’s Perspective
The subject of extractables for single-use bioprocess contact materials has been a subject of heated debate since roughly the summer of 2012, when the first ISPE paper was published issuing a call to action to develop a standardized extractables protocol for the industry (1). As a supplier that pioneered the science of extractables (2‒11) and has published extractables data for our products for over 20 years, Sartorius Stedim Biotech (SSB) took the opportunity to look back, take stock, rationalize, and…
A Product–Packaging Interaction Study to Support Drug Product Development
Drug packaging is subject to a number of regulatory requirements, including those for product containers and packaging. For example, according to the federal Food, Drug, and Cosmetic Act (FD&C) section 501(a)(3), a drug is considered adulterated “if its container is composed, in whole or in part, of any poisonous or deleterious substance which may render the contents injurious to health.” And 21 CFR states that drug packaging “shall not be reactive, additive, or absorptive so as to alter the safety,…