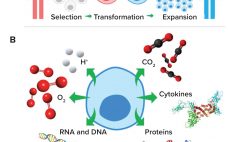
Chimeric antigen receptor (CAR) T cells first entered US clinics in 2017 (1), and this therapeutic modality holds tremendous potential as one of the most effective forms of personalized cancer care ever to reach patients. The revolutionary impact of CAR T-cell therapy comes from its ability to rewire our own immune defenses to kill cancer cells: It essentially modifies a patient’s naturally existing immune cells to boost their recognition and attack of cancer cells so that the person’s own immune system…
Cell/Gene Therapies
Innovations in the Manufacturing of Cell and Gene Therapies
Advancements in next-generation cell and gene therapies are fulfilling the promise of personalized medicine and attempting to cure and heal patients. Multiple approved products have been launched in global markets and the number of clinical trials continues to grow. Developing innovative advanced therapies is one of the biopharmaceutical industry’s greatest opportunities to dramatically improve patients’ lives. WuXi Advanced Therapies is a contract testing, development, and manufacturing organization (CTDMO) that has launched several innovative world-class platforms. It offers integrated platforms to…
Technology to Transform AAV Manufacture
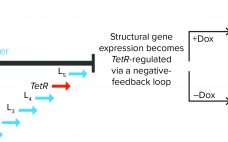
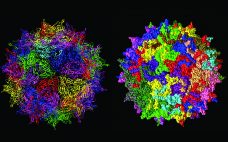
Adenoassociated virus (AAV) vectors are a popular choice for modern gene therapies because of their favorable safety profile, low immunogenicity, and the ease with which they can be transduced into different cell and tissue types. An AAV genome is a single strand of DNA comprising a replication (rep) gene, which encodes regulatory proteins involved in genome replication, and a capsid (cap) gene, which produces three capsid proteins. However, AAVs cannot replicate alone. In nature, AAV shares an exquisite relationship with…
eBook: Viral Vector Purification — A Discussion of Current Challenges and Methods
Adenoassociated viral (AAV) vectors have become synonymous with gene therapy delivery. However, because they are produced in such small quantities and because their upstream processes carry comparatively large amounts of host-cell DNA and other impurities, AAV purification can be challenging. Several researchers have applied different chromatographic strategies, but no universal method has been adopted in the biopharmaceutical industry. This eBook features a discussion among several industry experts that explores challenges specific to AAV purification, shedding light on whether current strategies…
eBook: Trends in Facility Design — In-House Manufacturing Considerations for Cell and Gene Therapy Production
Manufacturing and facility challenges facing cell and gene therapy companies are similar to but more complex than those encountered by companies that produce traditional biopharmaceuticals such as vaccines, monoclonal antibodies, and other therapeutic proteins. A single product can have multiple components, manufacturing of which may or may not be outsourced. Project timelines are short, production technologies are new and evolving, and clinical demands change rapidly. Increasing competition for contract manufacturing services requires reserving capacity far in advance, which in most…
Developing Advanced-Therapy Products Through Global CDMOs
Tremendous growth in the cell and gene therapy (CGT) industry is driving unprecedented demand for manufacturing services. To be sure, advanced-therapy developers increasingly are choosing to install in-house capabilities. Doing so can offer companies greater control of their processes, timelines, and budgets than they might have when outsourcing products (1). But industry experts agree that contract development and manufacturing organizations (CDMOs) will remain integral to CGT manufacturing and commercialization (1, 2), especially with veteran contract partners scrambling to acquire CGT…
The Role of New Technology in Enabling Cell and Gene Therapies
Moderator Doug Miller, with Tim Kelly, Daniella Kranjac, Andy Ramelmeier, and Ran Zheng This part of the BPI Theater program focused on emerging bioprocess technologies and their implications for cell and gene therapy (CGT) products. Doug Miller assembled a panel showcasing a wide range of technical expertise and representing several CGT industry stakeholders. That pluralism yielded far-reaching discussions about what CGT capabilities exist, what advances could facilitate therapy manufacture, and how companies might pursue technological development. Viral-Vector Therapies Miller kicked…
Viral-Vectored Gene Therapies: Harnessing Their Potential Through Scalable, Reproducible Manufacturing Processes
We might not associate the jazz queen Ella Fitzgerald with 21st-century gene-based therapies, but the First Lady of Song was on to something back in 1939 when she sang “’T’Ain’t What You Do (It’s the Way That You Do It).” Although demonstrating the safety and efficacy of gene-based therapies in rigorous clinical trials is essential for gaining product approval from regulators, doing the bare minimum is insufficient. The way that such products are produced also matters. Manufacturing processes and protocols…
Fluorescent Nanosensors: Real-Time Biochemical Measurement for Cell and Gene Therapies
Cell and gene therapies are destined to transform the methods by which global healthcare challenges are approached and overcome (1). The US Food and Drug Administration is reviewing and approving an increasing number of cell and gene therapy products (2), and biopharmaceutical developers are dedicating immense resources to realizing the enormous potential of these therapeutics. Therefore, technologies that facilitate their effective and efficient manufacture will accelerate cell and gene therapies’ transition from medicines of the future to medicines of the…
Gene Therapy Trends and Future Prospects
Gene therapy is defined as the transfer of genetic information to a patient for treatment of a disease. Clinical investigation of such therapies began in 1990 with a treatment for a rare immunodeficiency disorder and since has expanded to almost 1,000 clinical studies in 2019 (1, 2). In its most straightforward incarnation, the goal of gene therapy for genetic diseases is long-term expression of a transferred gene at levels that are high enough to be therapeutic, an approach sometimes called…