Speed to clinic testing — and then speed to market — are highly significant metrics for companies developing biopharmaceuticals. By increasing the pace of drug development, these companies can reduce costs, obtain revenues early, and establish commanding positions in the market relative to their competitors. High-throughput development tools have contributed much to the acceleration of drug development in recent years. Such technologies enable the testing of many process parameters in parallel. Combining them with multifactorial “design of experiment” (DoE) analysis…
Emerging Therapeutics
Antibody Derivatives: Deconstructing MAbs for the Next Wave of Biotherapies
Although they make up the largest and most successful category of biopharmaceuticals so far, monoclonal antibodies (MAbs) suffer from certain disadvantages. Some companies are addressing those limitations by deconstructing MAb molecules to create new emergent therapeutics. These antibody derivatives include: antibody fusions and fragments, bispecifics, trifunctional antibodies, and more. This eBook combines market analysis from consultant David Orchard-Webb with technical discussion from BPI cofounder and senior technical editor Cheryl Scott. It also includes commentary from editorial advisor Michiel Ultee and…
Cost of Goods Is Crucial for the Future of Regenerative Medicine: CAR-T Cell Therapy Provides a Case Study in Perspective
In the history of regenerative medicine, 2017 was a critical year. With approvals for Kymriah (tisagenlecleucel) from Novartis AG, Yescarta (axicabtagene ciloleucel) from Kite Pharma (a Gilead company), and Luxturna (voretigene neparvovec-rzyl) from Spark Therapeutics, cell and gene therapies finally made their mark on the regulatory landscape. Then in 2018, those products began both treating patients and bringing in revenues for their sponsor companies. “Patients are being treated, and biotechnology and pharmaceutical companies are being paid for treating them,” said…
Apparent Matrix Effects in an Iduronate 2-Sulfatase Specific Activity Assay
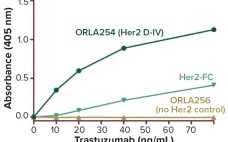
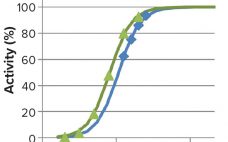
The recombinant fusion protein SHP631 consists of a chimeric monoclonal antibody binding to human insulin receptor and iduronate-2-sulfatase (I2S). This product is being developed as an enzyme replacement therapy to treat cognitive symptoms of Hunter’s syndrome. Because the current therapy (idursulfase, brand name Elaprase from Shire) cannot cross the blood–brain barrier (BBB), SHP631 is being developed to do so, enabling the presence of I2S in the brain. The enzymatic activity of this molecule is measured using the substrate 4-methyl umbelliferyl-α-L-idopyranosiduronic…
Partnerships in Immunotherapy for the Future of Cancer Treatment
Immunotherapy seeks to harness the power of our human immune system to fight disease. In this rapidly evolving field, collaboration among different stakeholders is essential to bringing new treatments to market. Patient advocacy groups, researchers, hospitals, manufacturers, and government entities all are working together to translate promising new research into life-saving products. Types of immunotherapy include monoclonal antibodies (MAbs) and antibody derivatives, checkpoint inhibitors (immune-modulating proteins), cancer vaccines, T-cell therapies, and cytokines — so the approach involves a range of…
A Brief History of Adherent Cell Culture: Where We Come From and Where We Should Go
In the past 20 years, novel therapeutics have become a major segment of biopharmaceutical research and development, particularly for immune disorders and cancer. Progress in gene therapies could bring cures for once deadly and debilitating genetic disorders such as hemophilia or muscular dystrophy. Biologic drug products offer potential treatments that have not been possible with traditional (chemistry-based) approaches. But such products also are more difficult to produce cost effectively at an industrial scale because of the intricacies associated with biological…
Messenger RNA Drugs: Engaging the Machinery of Patients’ Cells to Therapeutic Effect
Although most of the bioprocess industry has focused on process development for large-molecule formulations (e.g., protein drugs), a growing segment of the industry has been concentrated on other types of biotherapeutics to leverage advances in understanding of immunology and genetic engineering. Such technologies may emerge both as tools for drug manufacturing and at some point, as biopharmaceuticals, biotherapeutics, vaccines, and cell and gene therapies, themselves. What brings mRNA research to BioProcess International’s attention is the increasing interest turned toward therapeutic…
Development and Biomanufacturing Strategies for Next-Generation Antibody-Drug Conjugates
The development and manufacture of antibody-drug conjugates (ADCs) requires a series of complex steps. ADC manufacturers must comply with guidelines for both the small-molecule linker-drug and the monoclonal antibody (MAb). The authors describe their company’s development of its lead ADC product. They review process decisions, including the issues that factored into their selection of single-use systems, manufacturing challenges (differences between ADCs and MAbs), and testing methodologies for extractables and leachables. Synthon began as a small-molecule generics company in 1991. During…
Rolling with the ‘Tides: Elucidating the Role of Peptides and Oligonucleotides in the Biopharmaceutical Industry
In earlier issues of BPI we published a few “Elucidation” closers that we called “Defining Moments.” Since then, we have tried to distinguish key confusable terms from one another. Those presented (and sometimes “elucidated”) have been analytical and bioanalytical, spectroscopy and spectrometry, and biosimilars and biobetters. They are just a few of the many confusable terms in the biopharmaceutical industry. For example, when someone says “drug delivery,” a formulator will think of a syringe or transdermal patch, but a logistics…
Partnerships in Immunotherapy: Working Together to Take Cancer Treatment to the Next Level
Biopharmaceuticals are a particularly complex expression of medicine — and immunotherapies perhaps even more so. As treatments, these products themselves often also need “partners” of a kind: e.g., radiation/radiotherapies, traditional MAbs, and chemotherapies. Just as this field of endeavor requires the input and expertise of many different disciplines — from medical researchers to process engineers, clinicians to business leaders, and market experts to policy makers — this discussion of the topic of partnerships in immunotherapy brings together different experts in…