Avid Bioservices is a contract development and manufacturing organization (CDMO) specializing in mammalian cell culture process development and CGMP production of clinical and commercial-scale monoclonal antibodies, recombinant proteins, and enzymes. Committed to the success of our clients, our team constantly strives to build partnerships that extend well beyond the delivery of your product. By combining our knowledge and experience with a flexible and efficient approach, we can meet your specific project requirements Established CDMO with Proven Capabilities
Search Results for: antibody characterization
Smart Biologics Development and Manufacturing, and SMARTag™ Technology: For ADCs, Bioconjugates, and ADVASEPT® Aseptic Filling
From drug and biologic development to delivery technologies and supply solutions, Catalent is the catalyst for your success. With 80+ years of experience, we have the deepest expertise, broadest offerings, and the most innovative technologies to help you get more molecules to market faster, enhance product performance, and provide superior, reliable manufacturing and improved results. Catalent is your strategic partner for biologic drug development and manufacturing success. SMARTag™ technology offers a new toolkit to optimize antibody–drug conjugates (ADCs) and bioconjugates.…
Rapid Affinity and IgG Titer Analytics: Automating Bioanalysis with Gyrolab Nanoliter-Scale Technology
In the development of biotherapeutics, there is a need to quantitate antibody titer and to characterize antibody affinity against the target of interest. Such analyses are often challenged by the need for sensitivity, reproducibility, and higher throughput. Gyrolab systems automate and miniaturize affinity measurements and IgG titer quantification in a single platform using only nanoliter volumes with rapid time to results. IgG Titer Immunoassays with Broader Ranges for Cell Line Development
Tosoh Bioscience Introduces TSKgel® Protein A-5PW HPLC Columns
Tosoh Bioscience LLC, a provider of chromatographic solutions for the separation of biomolecules, today announced the introduction of the TSKgel Protein A-5PW column. This column is specifically designed for the rapid separation and robust monoclonal antibody titer quantification during cell line selection or upstream bioprocess optimization and control. The TSKgel Protein A-5PW column is now available for sale in the Americas from Tosoh Bioscience LLC. The TSKgel Protein A-5PW column is packed with 20 μm hydroxylated methacrylic polymer beads coupled…
Using Technology to Overcome Bioprocessing Complexity: Advanced Concentration and Analytical Technologies Accelerate Development and Manufacture of mAbs, Vaccines, and Biosimilars
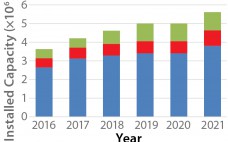
Unlike chemically synthesized drugs, whose structure is known and reproducible, biological drugs are derived from living cells and are sensitive, complex mixtures requiring cutting-edge biological technologies for their production. The growing importance of biosimilars in recent years is reflected in a corresponding rise in market value. The value of the global biologic therapeutic drug market reached approximately US$230 billion in 2014 and, according to BCC Research, will increase to nearly $390 billion by the end of 2019. This corresponds to…
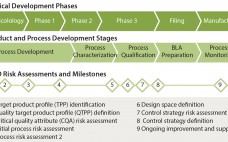
Quality By Design for Monoclonal Antibodies, Part 1: Establishing the Foundations for Process Development
The quality by design (QbD) modernized approach to pharmaceutical development is intended to provide regulatory flexibility, increased development and manufacturing efficiency, and greater room to innovate as well as improve manufacturing processes within defined ranges without obtaining regulatory approval first. QbD is a systematic developmental approach that starts with a clear goal in mind and emphasizes understanding of how variability in both process and materials affects a final product (1). Historically, product quality has been assured either with end-product testing…
From CMO to CDMO: Opportunities for Specializing and Innovation
Biopharmaceutical contract manufacturing organizations (CMOs) were initially enabled when the requirement for a company to file for both an establishment license application and a product licensing application transitioned to the current format of a biologics license application (BLA) submission for biological products (1). The initial focus of such CMOs was to provide large-scale, commercial manufacturing for companies that had already developed and validated bio manufacturing processes. Consequently, CMOs were generally formed as stand-alone service providers that “rented” manufacturing capacity to…
Outsourcing Biosimilars Development
A rapid increase in the number of companies working on development and registration of biosimilars has created a significant market for contract testing and manufacturing organizations (CTOs and CMOs) providing outsourced services specific to these products. Biosimilar developers turn to contract organizations when they lack either the internal capability or capacity for conducting certain work as well as when they require additional resources to bring products to market rapidly. A wide range of contract services are available, and each particular…
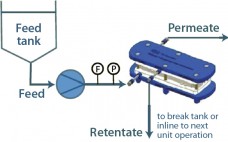
Virus-Filtration Process Development Optimization: The Key to a More Efficient and Cost-Effective Step
Size-exclusion–based parvovirus filtration is an important step toward drug product safety in biopharmaceutical production. However, once a virus filter is in place, and the required virus safety is ensured, less attention typically is paid to its optimization within the process. That might seem odd given that virus filtration can be one of the more expensive downstream processing steps ($/g protein processed). Most likely, the lack of attention can be attributed to aggressive timelines, limited process development resources, and the virus…
Alkyl Mono- and Diglucosides: Highly Effective, Nonionic Surfactant Replacements for Polysorbates in Biotherapeutics — a Review
Many biotherapeutic proteins are naturally subject to aggregation. The clinical consequences of protein aggregation can be dramatic, not only affecting bioavailability and pharmacokinetics, but in extreme cases dramatically altering pharmacodynamics as well. Of equal or perhaps more importance is that aggregation is a principal source of unwanted immunogenicity in biotherapeutics. Aggregation-induced neutralizing antibodies and/or anaphylactic reactions are serious and growing US and European regulatory concerns. So they will have significant and growing influence on the future development and regulatory approval…