The COVID-19 novel coronavirus pandemic has highlighted the biopharmaceutical industry’s need to create and implement chemistry, manufacturing, and controls (CMC) strategies that can expedite drug development during times of crisis. This bold topic was the premise of the 2021 CASSS Well-Characterized Biotechnology Products (WCBP) forum titled “Special Edition: Creating CMC Strategies for Pandemics and Beyond.” Held virtually on 25–28 January and 1–4 February 2021, the eight-day event addressed manifold considerations for SARS-CoV-2 virus prevention, diagnosis, and treatment. Five hundred attendees…
Business
A Practical Approach to Clinical Trial Diversity
A 2021 study in the Journal of the American Medical Association found that minority groups were underrepresented in US-based vaccine clinical trials, with white people accounting for ~80% of participants (1). Although researchers broadly acknowledge that trial representation should mirror the many different populations who could benefit from a drug, lack of diversity continues to hamper clinical-trial effectiveness. To address this problem, the pharmaceutical industry must increase awareness about clinical trials and reduce barriers to participation in them. Benefits of…
eBook: Speed to IND — Practical Wisdom for Uncertain Times
Even in “normal” times, companies need to balance time to filing an investigational new drug (IND) application against careful consideration of processes that can have far-reaching consequences on the quality of biologic products. But supply-chain interruptions still feature prominently in the news during the ongoing COVID-19 pandemic. From equipment to chemicals to plastic components, end users, their suppliers, and (critically) their suppliers’ suppliers all are feeling growing uncertainty about production timelines and availability of materials. In this eBook, BPI’s editor…
Collaboration Agreements: Critical Issues and Common Pitfalls
The number of collaboration arrangements for emerging technologies is increasing significantly, especially among companies developing COVID-19 diagnostics, vaccines, and therapeutics. Consequently, a massive amount of capital was invested in the biopharmaceutical industry in 2020. These deals range in value from the low millions to several billion dollars, representing significant risks and value for investors. Some recipients of that capital are seeking partners with whom to collaborate, sharing both the costs and risks associated with their activities and bringing innovative technologies…
Implementation of Established Conditions: Learnings from a “Sharing Science Solutions” Workshop
The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guideline Q12 (1) (step 4 sign-off in November 2019) is in the process of being implemented in a number of regulatory regions. The document provides additional frameworks for pharmaceutical life-cycle management. It is intended to support globally harmonized regulatory tools such as established conditions (ECs) and product life cycle management (PLCM) documents to facilitate postapproval changes to chemistry, manufacturing, and controls (CMC). Although a harmonized framework…
Intellectual Property and COVID-19
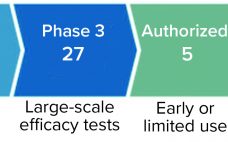
The COVID-19 pandemic is the greatest global health threat caused by a virus since the influenza pandemic of 1918 (and, before that, innumerable smallpox outbreaks throughout history). So far, the number of infections and deaths has not reached levels seen during the “Spanish flu” pandemic. However, travel, global trade, and modern factors such as misinformation on social media have increased infection rates and risks of infection. As of 15 July 2021, the World Health Organization (WHO) had reported 188,655,968 confirmed…
Risk Determination of Potential Mycotoxin Exposure to Patients: Testing Recombinant Human Factor VII from Transgenic Rabbits
Sevenfact eptacog beta is a new recombinant human factor VIIa (rFVIIa) developed by LFB SA in Les Ulis, France, as a bypassing agent (BPA) for treatment and control of bleeding in people with hemophilia A and B and inhibitors (1, 2). The product was approved for use in adults and adolescents by the US Food and Drug Administration (FDA) in April 2020 (3). It is expressed in the milk of transgenic rabbits and purified through a multistep process using both…
Focusing on the Patient Journey Can Increase Access to Lifesaving Therapies
Cell and gene therapies (CGTs) are positioned currently as last-chance, “miracle” cures for patients who have severe illnesses. Such promises require innovation. Despite the cutting-edge science and significant investment that goes into CGT development, fundamental challenges remain, including patient access. The highly personalized nature of autologous-therapy development presents myriad logistical, financial, and manufacturing challenges to ensuring global access to treatment. Understanding a patient’s journey to treatment is vitally important to achieving that goal. Barriers to Cell and Gene Therapy Access…
eBook: Rare Diseases — Biopharmaceutical Challenges Presented By Relatively Small Patient Populations
By definition, an orphan disease affects a small percentage of the population. However, with some 7,000 such conditions identified so far, they collectively have a significant impact on global health. An estimated 350 million people are affected worldwide by a rare disease — altogether more than the population of the world’s third largest country (the United States). Some well-known biopharmaceutical companies are devoted to developing treatments for rare diseases. However, the vast majority of such diseases have no treatments approved by…
Preparing for a Virtual Audit
Audits are a vital quality-management tool of the biopharmaceutical industry. Whether the objective is to verify supplier or partner qualifications, contribute to corrective and preventative actions (CAPA), or fulfill regulatory compliance requirements, conducting proactive auditing is key to successful operations. Over the past year, virtual audits —also known as remote or distance audits — have enabled biopharmaceutical companies to meet compliance and quality assurance demands despite COVID-19–related travel restrictions and social-distancing protocols. Notwithstanding the challenges of virtual audits, the benefits…