Process intensification offers many benefits but implementing PI can introduce an unexpected challenge of managing larger media volumes. This whitepaper helps ensure your implementation strategy accounts for the increased media volume associated with process intensification. What You Will Learn Gain the tools and knowledge needed to confidently assess intensification options with a focus on media management for new or existing facilities. Follow the media journey from prep to use, exploring potential logistical pitfalls in the management of increased media volumes…
BPI White Papers
A Frost & Sullivan Virtual Think Tank: 3M™ Polisher ST Beta Testing Series
One step can change everything: Replace your flow-through AEX column with a novel single-use polishing solution 3M™ Polisher ST single use polishing solution – from lab to manufacturing scale Frost & Sullivan recently invited a panel of bioprocessing industry leaders and key opinion leaders to participate in a Virtual Think Tank (VTT) Early Access series – a new and unique thought leadership forum. Each VTT panel, comprised of professionals from top pharmaceutical companies, contract development and manufacturing organizations (CDMOs) and…
Build vs. Buy: A Critical Calculation for Cell Therapy Innovators
Cell therapy is proving to be one of the most promising advanced modalities, representing a significant step forward in the treatment of a wide range of challenging diseases and conditions. As a cell therapy candidate advances from discovery through clinical development and ultimately to commercialization, foundational decisions must be made by the product sponsor that will impact both scientific and commercial success. One of the biggest decisions is whether to build a manufacturing facility or outsource to a contract development…
Five Ways the Amersham ImageQuant™ 800 GxP Helps You Remain Compliant
In highly regulated environments, such as the pharmaceutical industry, remaining GxP compliant while efficiently delivering your products to a competitive market is a challenging task, particularly when Western blot imaging is involved. While this can often be a tricky and time-consuming task, our Amersham ImageQuant™ 800 GxP imaging system and software streamlines the process. It provides simple solutions that support data traceability, accountability, and integrity. Confident decision making and GxP compliance go hand in hand with high-quality imaging. In this…
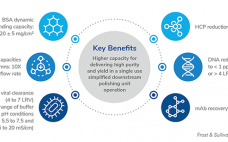
3M™ Polisher ST — The Next Frontier in Downstream Processing, a Frost & Sullivan White Paper
Advancements in next-generation therapies—Paving the way for novel bioprocessing solutions. Learn about 3M Polisher ST technology which utilizes a guanidinium-functionalized polyamide membrane protected by a Q functionalized non-woven material. This Frost & Sullivan whitepaper explains how the technology demonstrates the viability of replacing the depth filtration and anion exchange chromatography (AEX) steps to achieve a simplified and cost-effective process. The platform is designed to reduce process and product related impurities and offer robust performance across a wide range of process…
Scale-Down Optimization to Scale Up Success
As immune cell therapy advances to address new indications, the need for rapid development of robust manufacturing processes becomes increasingly important. Early process optimization sets the stage for clinical and commercial manufacturing and plays a foundational role in decreasing time to market and lowering COGS. This application note describes the streamlining of T-cell expansion optimization using a DOE-based approach in a semi-automated, controlled multi-parallel setup of the Sartorius T-Cell Exploration and Characterization Solution. Download to learn about: Rapidly screening media…
MAb Quantitation: Protein A HPLC vs. Protein A Bio-Layer Interferometry
Rapid, accurate and cost-effective quantitation of monoclonal antibodies (MAbs) is essential for bioprocessing. High Pressure Liquid Chromatography (HPLC) and the Octet® are some of the commonly used techniques for MAbs titer determination. To ensure MAbs purification column efficiency, the dynamic binding capacity (DBC) of Protein A for Mab can be determined by loading feedstock onto the column until binding sites for the MAb become saturated and MAb begins to break through. An assessment of the relative merits of Protein A…
Why Choose Eurofins BioPharma Product Testing?
For all stages throughout the development, manufacturing and release of your biological product, Eurofins BioPharma Product Testing offers comprehensive, fully CGMP-compliant Viral Clearance Services. We have two dedicated clearance suites for extensive capacity and timely execution and reporting of your study results. And all of our assays are fully validated to meet ICH Q2 requirements. Why Choose Eurofins BioPharma Product Testing? We bring together leading experts in the industry with extensive scientific and regulatory experience. By supporting you from study…
The Viscosity Reduction Platform: Viscosity-Reducing Excipients for Protein Formulation
Protein viscosity is one of the major obstacles in preparing highly concentrated protein formulations suitable for subcutaneous (subQ) injection. Highly viscous protein solutions would require a significant force to be applied to the syringe for injection. As a result, the patient could experience a considerable amount of pain. In many cases, injectability would not be possible. When characterizing protein viscosity behavior, one can differentiate two different concentration regimes. At very low concentrations below about 75 mg/mL, proteins are rarely viscous.…
Accelerating Discoveries for Viral Biology and Host Immunity with Advanced Cell Analysis Solutions
This whitepaper summaries recently published studies that incorporated the Incucyte® Live-Cell analysis platform and iQue® 3 Advanced High-Throughput Flow Cytometry. These state-of-the-art technologies were used to simplify workflows and provide rapid analysis solutions and enhanced throughput, aiding the discovery of new insights on the host-pathogen life cycle throughout the course of infection. Post-infection, these platforms were leveraged to monitor exposure, assess immune protection, and accelerate the development of anti-viral small molecules, neutralizing antibodies, and vaccines. The Incucyte® platform offers automated…