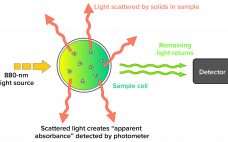
Turbidity describes the relative clarity of a liquid as the result of suspended solids. Instruments that measure turbidity typically use a beam of light to detect particles by measuring the difference between the amount of light emitted from the light source and the amount that is received by a detector. Such measurements are affected by the size, shape, and number of particles in a sample of liquid because those solids scatter the incoming light, which provides an apparent absorbance that…
Analytical
The Five Heresies of Cell Culture: Debunking Conventional Wisdom
Cell culture and bioprocessing conventional wisdom remains a hurdle for the wider adoption of more precise tools. It has been more than 60 years since any real progress has been made towards creating a more accurate and reliable way of performing cell culture monitoring to better understand the effects of things like pH and oxygen at the pericellular level. At SBI, we’re developing optical sensing technologies that unlock the “black box” of cell culture to bring actionable insights to scientists…
Cell-Line Development: Accelerating Antibody Discovery By Monitoring Titer and Glycosylation with the Octet Platform
Cell line development involves the screening of thousands of clones in an effort to find the few optimal clones that are stable, grow as expected, and produce high yields of the bioproduct. The time it takes from engineering an optimal cell line to the production of the target biologic can be prohibitive and may differ from molecule to molecule. While expression-level analysis like titer screening is carried out early, other critical quality attributes (CQAs) such as glycan characterization are often…
Measuring Cell Density in HyPerforma S.U.B.s with ABER Futura neotf
Single-Use Sensors
Monitoring critical process parameters (CPPs) and key performance indicators in bioreactor control systems is crucial to ensure proper cell growth and protein production. Today, most of the major biopharmaceutical companies employ capacitance measurement, in R&D and through process development to manufacturing. Owing to the increased use of single-use bioreactors and building on Aber’s experience with single-use capacitance sensors, the latest Futura neotf single-use capacitance sensors have been specifically developed for integration into Thermo Fisher Scientific bioprocess containers (BPCs) for use…
Cell Banking in the Spotlight: Advising Biologics Developers About Cell Bank Preparation and Characterization
Living cells are at the heart of biotechnology, and cell lines for production and testing of biopharmaceuticals are highly valuable assets. The process of banking cells generally moves from development of a research cell bank (RCB) based on a clone of interest to establishment of a master cell bank (MCB), from which working cell banks (WCBs) can be produced. Especially for biotechnology startups, preparation of an MCB can involve a significant jump from work performed in standard laboratory conditions to…
Ask the Expert: Considerations for Mass Spectrometric HCP Analysis During Process Development
Host-cell proteins (HCPs) must be assessed early in biopharmaceutical product development to establish the efficiency of a purification process. Liquid chromatography coupled with mass spectrometry (LC-MS) is a strong orthogonal method for HCP identification and quantitation. During an 18 March 2021 “Ask the Expert” presentation, Christina Morris (senior scientist at BioPharmaSpec) explained how MS analyses can enhance process development (PD), then presented considerations for generating spectral libraries, selecting samples for quantitation, and reviewing data. Morris’s Presentation Spectral Libraries: A typical…
Expanding the Flow Cytometry Toolkit with Next-Generation Polymer Dyes
Since the emergence of SARS-CoV-2, scientists have discovered more about the role of our immune systems and cytokine-associated processes responsible for systemic immune reactions that are typical in patients with COVID-19 (1). Flow cytometry is used to understand those processes at a single-cell level. It is the standard method used in immunology to characterize multiple phenotypic and functional parameters of single cells, including cytokine analysis. Despite advances in flow cytometry instrumentation, reagents, and analytical software, several challenges remain. Conjugated antibodies…
eBook: Bioreactor Sensors —
Inside the Dynamics of Cell Culture
Cell culture monitoring can fall into something like a “black box” conundrum. Efforts to measure key parameters such as pH, glucose, and even cell density require sampling and removal of the contents from a bioreactor. But that procedure can expose both a process and an operator to contamination risks. Emerging bioreactor sensors are designed to address some of those challenges, but the rapid adoption of single-use technologies and the rise of perfusion cell culture have presented obstacles to their implementation.…
How Capacitance Measurement Can Improve Viral Vector and Virus-Based Vaccine Production
With the increasing development of viral vector and virus-based vaccines, technologies that help to manufacture and scale up these types of vaccine quickly and cost-effectively have become more critical. By accessing this special report from Aditya Bhat, a capacitance technology expert at Aber Instruments, you will find out how in-line capacitance measurement can produce a detailed fingerprint of cell culture processes and how that can benefit vaccine production. From case studies involving baculovirus, AAV, and measles, you will discover why…
Virus Assay Variation Is the Main Source of Variation in Viral Clearance Studies: Retrospective Analysis of a Large Data Set
Biopharmaceuticals produced from mammalian cell cultures are susceptible to viral contamination. That risk is mitigated by applying complementary approaches. Those include extensive testing of cell banks, selecting low-risk raw materials, testing cultivations for viruses, and documenting the capacity of a purification process to inactivate and remove viral contaminants. The latter commonly is referred to as viral clearance and usually expressed as a log reduction value (LRV). Novo Nordisk has performed several viral clearance studies for different processes and process steps.…