The boundaries of technology can be pushed significantly when insights from different fields reinforce each other. Based on in silico simulations demonstrating the importance of order on the efficiency of chromatographic separations, PharmaFluidics has combined expertise from the analytical chromatography and semiconductor chip manufacturing industries to create a new type of nanoscale liquid chromatography (LC) column. Conventional LC columns contain randomly packed beads as a stationary phase. By contrast, PharmaFluidics uses a lithographic etching process to create a perfectly ordered…
Downstream Processing
Scaling Considerations to Maximize the High-Area Advantage
Maximizing filtration-area density is a design strategy to minimize filter footprint and improve filtration process economics. Pleated membrane formats commonly are used to achieve that goal for sterilizing-grade filters operating in dead-end mode (also known as normal-flow filtration). Although high-density pleat geometries increase productivity for a device, such formats can present unique challenges. One of the most common concerns is that pleat formats can introduce flow resistance that impedes a device’s filtration efficiency, particularly for high–pleat-density geometries (1, 2). Filtration…
Current Thinking in Viral Safety: Risk Management Protects Patients
BPI’s editor in chief S. Anne Montgomery recently caught up with long-time editorial advisor Hazel Aranha (purification technologies technology expert for Sartorius Stedim Biotech, North America). They discussed a number of topics related to viral safety. Montgomery: What is the current thinking regarding virus-safety assurance in biopharmaceutical manufacturing? How is the industry preventing viral contamination? Aranha: The “holy grail” of viral safety — absolute freedom from extraneous agents or residual pathogenicity — is a myth. That said, biopharmaceutical products have…
Scalable Purification of Viral Vectors for Gene Therapy: An Appraisal of Downstream Processing Approaches
Gene therapy is the transfer of genetic material to a patient’s cells to achieve a therapeutic effect. Therapeutic DNA is largely delivered using viral vector systems based on adenoviruses (Ad), adenoassociated viruses (AAV), and lentiviruses (LV). With the application of such viral vectors as clinical therapeutics growing, scalable commercial processes (particularly for purification) are being investigated and optimized to best ensure that critical quality attributes (CQAs) are retained. Herein we review viral vector purification techniques and the effect of different…
A Statistical Approach to Assess and Justify Potential Product Specifications
As stated in ICH Q6B, specifications are critical quality standards that are both proposed and justified by drug product manufacturers. Xiaoyu et al. provide information on several statistically based strategies to establish specification acceptance criteria (SAC) (1). Here we address an alternative approach to relate proposed SAC for quantitative data to relevant lot history. In particular, proposed SAC can be derived in part by using calculated limits for which the lower bound of an approximate 95% confidence interval for the…
Examining Single-Use Harvest Clarification Options: A Case Study Comparing Depth-Filter Turbidities and Recoveries
Steadily increasing demand for biopharmaceutical drugs has led the industry to examine its manufacturing scales while pressuring research and development groups to produce high-yielding clones and processes. Improved media, feed supplements, bioreactor designs, and control of process parameters have helped biomanufacturers achieve multifold increases in volumetric productivity from production bioreactors. However, cell culture processes are significantly affected by their bioreactor’s ability to support cells at higher densities and sustain cultures at lower viabilities. With the implementation of a number of…
Downstream Disposables: The Latest Single-Use Solutions for Downstream Processing
Downstream processing has been considered a “bottleneck” in the manufacture of protein biotherapeutics ever since cell culture engineers began dramatically improving production efficiencies around the turn of the century. And as single-use technologies have grown in importance and acceptance, offering more solutions every year, their biggest challenges too have been in the separation, purification, and processing that follows product expression in cell culture. Many of the technologies familiar to process engineers — e.g., centrifugation and chromatography — present technical and…
Single-Use Depth Filters: Application in Clarifying Industrial Cell Cultures
For current process development phases, many biomanufacturers’ attention is directed increasingly to the first unit operation in downstream processing, which is the removal of cells and cell debris from culture broth and clarification of supernatant containing a biopharmaceutical product. Given the high cell densities achievable with both mammalian and microbial cell culture processes, primary recovery can be a significant challenge. The current trend in cell culture is to increase product titers with enriched culture media, improved cell productivity, and increased…
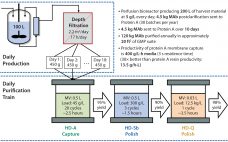
Multicolumn Chromatography: Facilitating the Commercialization of Monoclonal Antibodies
Since 2001, global contract development and manufacturing organization (CDMO) CMC Biologics has completed more than 120 projects with at least 100 pharmaceutical partners. During that time, the company has taken a holistic approach to helping clients balance manufacturing risks and rewards. The team focuses on evaluating key technologies to deploy a constantly evolving set of capabilities in support of biopharmaceutical clients throughout their product lifecycles. Part of that commitment is continually evaluating what would best benefit customers and where key…
Membrane Adsorbers, Columns: Single-Use Alternatives to Resin Chromatography
Filtration membranes are used extensively throughout the biopharmaceutical industry for a range of applications, from coarse filtration to nanofiltration. Advantages of filter technologies include easy scaling, disposability, and (for many membrane filters) rapid and robust performance in a single-pass. The same advantages have been realized with membrane adsorbers. Chromatography resins are inherently disadvantaged by diffusion limits of the pores in chromatography media. Therefore, resin columns must be significantly oversized to match the performance of high productivity bioreactors. By comparison, membrane…