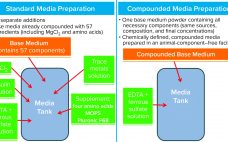
Cell culture medium is critical to cell growth, metabolism, and protein expression. It provides for optimum pH, osmolality, and nutrients in an environment that is essential for cell survival, growth, and expression of proteins and/or metabolites and drug-substance modalities of interest (1). A complete medium typically contains basic nutrients such as carbohydrates, amino acids, lipids, salts, vitamins, trace metals, growth factors/hormones (e.g., insulin), antishear factors, and other chemicals that facilitate cell growth and protein expression and may stabilize recombinant protein…
Biochemicals/Raw Materials
Fetal Bovine Serum Source Countries: Comparing Regulatory Animal Health Infrastructures
The motto of the European Serum Products Association (ESPA) — “Serum Saves Lives” — reaffirms the essential role that animal serum plays in cell-culture–based research and applications to protect the health of both human and animal populations. Animal serum, especially fetal bovine serum (FBS), needs to be available in abundant supply and at affordable prices to medical and veterinary facilities all over the world. With one billion cattle globally, the supply of FBS should be plentiful, but not all countries…
Intensified Seed Train Strategy for Faster, Cost-Effective Scale-Up of Biologics Manufacturing
The high costs of and limits on global accessibility of biologics such as monoclonal antibodies (MAbs) are focusing the biopharmaceutical industry’s attention on strategies for rapid, economical development of such therapies. Process intensification is one approach to help shorten manufacturing timelines and reduce cost of goods (CoG) (1, 2). Today, process intensification in upstream cell culture enables biologics manufacturing in facilities with smaller footprints and lower scale-up volumes than was possible before. Intensified processing of Chinese hamster ovary (CHO) clones…
Cryopreserved Leukapheresis Materials Help Alleviate Donor Sourcing Issues
Reliable access to high-quality starting material is a primary challenge in cell therapy manufacturing. Freshly isolated leukopak starting materials depend on donor availability and are vulnerable to cell losses from scheduling changes and other unforeseen circumstances. Flexibility often is required for cell therapy manufacturing. Therefore, relying solely on freshly isolated starting material is impractical, particularly when cell therapy logistics involve global shipping and distribution. Donor sourcing is among the most critical factors shaping cell and gene therapy (CGT) supply chains.…
eBook: Raw Material Control Strategy — Leveraging Knowledge of Material Attributes and Data Analytics as Key Elements
Ensuring pharmaceutical quality begins with in-depth understanding of process/platform capabilities, which is informed by knowledge gained through product and process development, subject-matter expertise, and lessons learned from experience. And all outside factors that can affect manufacturing outcomes must be taken into consideration. Extra vigilance is necessary for understanding potential sources of variation and maintaining robust control strategies to ensure process consistency — and ultimately product quality for patients. Biomanufacturing unit operations require multiple raw materials that must be documented as…
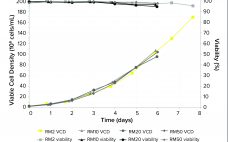
Using Peptones to Achieve Diverse and Demanding Bioproduction Goals
As bioproduction requirements advance, it is critical to have consistent, high-quality media and supplements that continue to meet evolving industry needs. Peptones have been successfully used in bioproduction applications for more than 30 years to meet diverse and demanding production requirements. Their unique nutritional profiles and usage flexibility make peptones ideal components for creating a robust bioprocess. This Special Report will demonstrate the benefits of peptones and how they can be used to enhance process performance and consistently yield a…
Toward a Roadmap for Cell-Free Synthesis in Bioprocessing
Cell-free synthesis (CFS), also known as cell-free transcription and translation, supplements cellular components (either a cell lysate or purified recombinant elements) with nucleotides, amino acids, metabolic intermediates, and salts to produce a nucleic acid or protein from a genetic template added to the reaction. This exciting technology has seen a substantial increase in both academic and commercial interest over the past decade (1). Interest stems in large part from the potential to democratize access to the machinery of biology by…
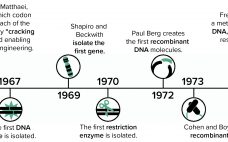
Microbial Expression and Purification: One Company’s Historical Perspective
Since the dawn of the recombinant DNA era in the 1970s, New England Biolabs (NEB) has been integrally involved in expressing and purifying proteins, both for its own research interests and for biomanufacturing processes. In 1978, the company began screening microorganisms for restriction enzymes. Our scientists remember the challenges met in purifying limited amounts of restriction enzymes and other proteins from native organisms isolated from the environment. The efforts of those scientists to clone, overexpress, and purify restriction enzymes from…
BVDV Risk Mitigation: Dealing with Bovine Viral Diarrhea Virus in Serum
Bovine serum products such as fetal bovine serum (FBS) are critical, nutrient-rich supplements frequently used in cell culture systems for a number of applications, including biotechnology, animal and human pharmaceutical and diagnostic manufacturing, and life-science research. Serum can be contaminated with adventitious agents that could increase its risk for use in cell culture systems. Bovine viral diarrhea virus (BVDV) is one of the most significant infectious diseases in the livestock industry worldwide because of its high prevalence, strong persistence, and…
Brazilian JBS Federally Inspected Fetal Bovine Serum
Serum is the most commonly used supplement in cell culture. Fetal bovine serum (FBS) is the common choice because it contains high concentrations of growth factors and other important signaling molecules (e.g., adhesion proteins, nutrients, carrier proteins, cytokines, and hormones) required for cell survival and differentiation together with its buffering capabilities. FBS production begins with the collection of whole blood from bovine fetuses under aseptic conditions. Once collected, the blood is allowed to clot, and the serum is mechanically separated.…