Oligonucleotide-based therapeutics have been studied over recent decades, and their promise as a new drug modality is now being realized. The growing interest in oligonucleotides is driven by their high potential for treating different medical conditions, the growing number of oligonucleotide drugs approved by the US Food and Drug Administration (FDA), an increased focus on personalized medicines, the development of therapies for rare diseases, and the wide adoption of nucleotide-based COVID-19 vaccines. Oligonucleotides are short, linear sequences of DNA or…
Nonantibody Biologics
eBook: mRNA — Revisiting a Technology That Has Rocketed into Success
At the end of 2018, BPI published its first eBook about mRNA drug products — and quite a lot has happened since then! Our initial report highlighted companies working on mRNA therapeutics for cystic fibrosis, heart disease, and cancer, as well as vaccines. The latter approach took off in 2020 with the advent of SARS-CoV-2 and the COVID-19 pandemic, and in a stunningly short time, the biopharmaceutical industry has learned much about manufacture, formulation, product design, and distribution of mRNA…
SARS-CoV-2 Hyper-Immunoglobulin: Purification and Characterization from Human Convalescent Plasma
The novel severe acute respiratory syndrome coronavirus 2 (SARS- CoV-2) emerged as a major pandemic coronavirus disease in 2019 (COVID-19) and since then has killed many people and paralyzed the global economy (1, 2). With specific antiviral therapeutic agents or antibodies yet to be approved, other antivirals and novel vaccine strategies have been essential to containing the virus and disease transmission. Passive antibody therapy can be used to limit the scope of epidemics by providing patients with antibodies that recognize…
Ask the Expert: Developing Strategic Analytical Programs for Therapeutic Peptides
Ashleigh Wake began her 15 October 2020 “Ask the Expert” presentation by pointing out that peptide products are manufactured in a “regulatory vacuum.” Peptide-product developers must be strategic in designing characterization and quality control (QC) programs. Wake reviewed available methods and explored key considerations for developing phase-appropriate analytical controls. Wake’s Presentation Because peptides overlap small- and large-molecule drugs in size, regulatory expectations differ by product size and clinical indication. Thus, analytical programs should be designed around critical quality attributes (CQAs)…
Removing Oligomers of a Recombinant Human Therapeutic Hormone:
Evaluation of Chromatographic Options for Effectiveness
Aggregation is a common cause of protein instability, which renders a biologic product unfit for therapeutic use. Sometimes it is difficult to purify monomeric proteins from oligomers because of similarities in their isoelectric points (pIs). Proteins such as hormones have pI ranges similar to their oligomers and thus can be difficult to separate out using a conventional polishing chromatographic step such as ion exchange. With those pI similarities, removal of oligomers to a considerable extent by ion exchangers can compromise…
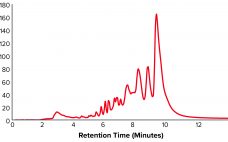
A Universal Assay Determination Method for Antisense Oligonucleotides: A New Slope Spectroscopy Method
Antisense oligonucleotides (ASOs) are short, synthetic, single-stranded oligodeoxynucleotides that can alter RNA and reduce, restore, or modify protein expression through several distinct mechanisms. ASO technology has become an important drug discovery platform for most major pharmaceutical companies. To date, six antisense drugs have been approved by regulatory agencies to treat diseases spanning viral infections, hyperlipidemias, and neurological diseases. More than 50 additional ASO drugs are in clinical trials. For an ASO drug product, an assay of its active pharmaceutical ingredient…
Peptide Therapies: Designing a Science-Led Strategic Quality Control Program
Peptide therapeutics are a unique class of pharmaceuticals. They can fall into one or more regulatory categories: as conventional chemical molecules, biological entities, or biosimilars. Developing a well–thought-out quality control (QC) strategy is key to meeting development milestones and complying with evolving regulatory requirements. Safety assessment is critical because slight changes in the structure, physicochemical properties, stability, and impurity profile of a peptide can provoke an adverse immune response. A robust analytical QC program should be conducted in laboratories that…
eBook: Challenges in Industrial Process Development of Exosome-Based Therapies: Characterizing and Managing Diversity
The traditional classification of extracellular vesicles (EVs) includes three types: exosomes, microvesicles, and apoptotic vesicles. Each type arises from a distinct origin and exhibits distinct characteristics. The problem is that their size ranges overlap and that the major surface proteins presented by exosomes also are present on the surfaces of microvesicles and apoptotic bodies. This makes it a challenge for process developers to identify the vesicle fraction that best serves a particular exosome therapy. Anion-exchange chromatography (AEC) can fractionate EVs…
Fighting Alzheimer’s Disease with a Breakthrough Peptide
Even more so than cancer, Alzheimer’s disease is one disease that many people fear greatly. The thought of falling prey to the inevitable desecration of our own minds is something that makes even the bravest among us shudder. If we’re robbed of our sense of who we really are, we imagine, then we are doomed to live our last days without the dignity that defines us and that we hold most dear. The ultimate horror of the condition is that…
eBook: Peptide Therapies — Small Biomolecules Address Big Health Problems
This crash course in the development of peptide therapies brings together two articles: one on product research and the other on manufacturing and regulatory concerns. In “Fighting Alzheimer’s Disease from Within: A Breakthrough Peptide,” freelance journalist Marc Davis highlights current developments in Alzheimer’s disease treatment, focusing specifically on a peptide approach from Canadian company NervGen. In “Making Peptides Work,” BPI’s senior technical editor adds perspective on developing and manufacturing peptide therapies from the Informa Connect community, with commentary from experts…