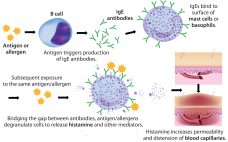
Rapidly increasing use of monoclonal antibodies (MAbs) in the treatment of neoplastic, autoimmune, and inflammatory diseases has led to a dramatic increase in hypersensitivity reactions worldwide, complicating the use of MAbs as first-line therapies and limiting patient survival and quality of life (1). The origins of anaphylaxis are not well understood, though its mechanism is fairly straightforward (Figure 1). It is usually attributed to some undefined intrinsic property or properties of a biotherapeutic — despite the fact that biotherapeutic formulations…
2017
Process Development of Microbial Plasmid DNA: Fast-Tracking with Modular Single-Use Minibioreactors
There has been a rapid rise in the number of positive clinical outputs from clinical studies based on gene and cell therapies. This is in addition to the licensing of products such as GlaxoSmithKline’s Strimvelis ex-vivo stem-cell therapy for treatment of severe combined immunodeficiency caused by adenosine deaminase deficiency (ADA-SCID) in 2016 (1) — has led to an increase in demand for therapeutic vector manufacturing capabilities. Viral vectors are being used for an increasing range of conditions, including monogenetic conditions.…
Insulin Improves Cell Growth and Influenza Yield in a HEK293 Suspension Cell Line
In her “Ask the Expert” webcast on 21 June 2017, Aziza Manceur of the National Research Council Canada (NRC Canada) discussed the benefits of using insulin in bioprocessing. Manceur’s Presentation Canada’s NRC has research facilities across Canada covering disciplines such as aerospace, energy, mining, and medical devices. The NRC serves both the government and the private sector, working with Canadian and international clients. Manceur is based in Montreal with the cell culture scale-up team (in the human health and therapeutic…
Emerging Markets: Current Insights into the State of Global Biopharmaceutical Manufacturing
Opportunities for establishing strong biopharmaceutical capabilities are expanding across the globe. This e-book seeks to encapsulate the current state of emerging markets/countries, tracing key elements above and offering examples to show where (in the world) the biopharmaceutical industry is expanding and securing its footholds. Generally, to succeed in these markets, foreign companies must exercise efficient resource management and control, show creativity and receptiveness to cultural differences, develop new strategies, and manage expectations. Working with local partners can provide access to…
Regulation, Analytical, and Process Issues with Leachables: Toward Harmonization for Latin America with Europe and North America
The pharmaceutical industry follows strict regulations regarding impurities, including process-related leachates. Plastic manufacturers use hydrophobic, nontoxic additives for manufacturing containers for use in the pharmaceutical and food industries. However, some issues about dealing with such impurities are not yet resolved. In developing countries, regulators are working on guidelines to help local companies ensure characterization of impurities. In this exclusive editorial eBook, authors from Mexico describe some issues related to plastic leachables in the context of ongoing efforts to harmonize regulations…
Push-Button Simplicity: Automatic Fermentation with the BioFlo® 120 Auto-Culture Mode
Learning to use a bioprocess controller is a complex endeavor. Even if someone has previous bioprocess experience, moving to a new software platform can entail much learning and reduce process efficiency. With the auto-culture modes of the Eppendorf BioFlo 120 bioprocess control station, a user can select with a push of a button either a predefined Escherichia coli batch fermentation protocol or a Chinese hamster ovary (CHO) batch cell culture process. Proof of Concept: E. coli Auto-Culture Fermentation The auto-culture…
Extractables and Leachables Studies: Designed and Performed to Meet All Intended Needs
There are no definitive requirements on how to perform extractables and leachables (E&L) studies. However, the US Pharmacopeial Convention recently released two chapters to provide guidance on performing E&L studies. USP chapters <1663> and <1664> discuss options and considerations in performing an extractables study and a leachables study, respectively. There are also various workgroups that have published guidance documents to assist industry in designing and performing these studies. The Product Quality Research Institute (PQRI), the Bio-Process Systems Alliance (BPSA), and…
Automation and Modularity Allow MAb Biotech to Cut Scale-Up Time
Original developers of biosolutions and products, especially those facing the debut of biosimilars in core markets, have an urgent imperative to reduce manufacturing costs through increased productivity and yields. In turn, this drives a wide range of business decisions, including capital investment, process choices and design, and equipment selection. To this end, biodevelopers are adopting more sophisticated processes (such as perfusion) to address low-titer cell lines and reduce raw material costs. They’re also seeking more sophisticated and flexible R&D and…
Looking into the Future: An Interview with Steve Bagshaw, CEO of Fujifilm Diosynth Biotechnologies
Steve Bagshaw, chief executive officer at FUJIFILM Diosynth Biotechnologies, offers some insight into the ever changing CDMO landscape and the future of medicine. Mr. Bagshaw is an industry thought leader and an advocate of the Biotechnology Industry. Q: Fujifilm Diosynth Biotechnologies has been expanding and making big announcements in the past year. Can you talk about how this growth is shaping your long-term vision as a contract development and manufacturing organization (CDMO)? We are seeing the benefits of long-term investment.…
Single-Use Diaphragm Valve
There is now a larger selection of the world’s first controllable single-use diaphragm valve: GEMÜ SUMONDO. GEMÜ, the leading manufacturer of valve designs for the pharmaceutical industry, has established the first controllable single-use diaphragm valve on the market: the GEMÜ SUMONDO. In addition to a pneumatically operated version, the product range now includes a version with a hand wheel for manual operation. And because of increased customer demand globally, the product range has been expanded in the area of associated…