Electrospray-ionization mass spectrometry (ESI-MS) is a well-established tool for biotherapeutic analysis. It draws intact proteins or peptide ions into the vacuum of a mass spectrometer, where the ion mass is measured. Electrospray ion-mobility mass spectrometry (ESI-IMS) introduces ions into a low-pressure gas, where the effects of aerodynamic drag reveal their shape. This technique is just emerging as a valuable tool for characterizing intact proteins, even though for a decade it’s been the basis of a commercially available medical diagnostic test…
Manufacturing
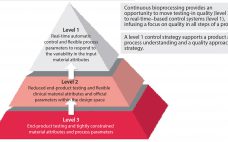
The Industry’s Hesitation to Adopt Continuous Bioprocessing: Recommendations for Deciding What, Where, and When to Implement
The US Food and Drug Administration has stated its appreciation of continuous bioprocessing (CBP), and some studies have shown that it can save manufacturers time and money. However, the bioprocessing industry is still reluctant to implement continuous bioprocessing right away. It will be interesting to see which companies will be among the first-movers to harness the competitive benefits. Although few biologics today are made using CBP-enabled equipment (e.g., advanced bioreactors), the industry is changing. For biologics already in production, it…
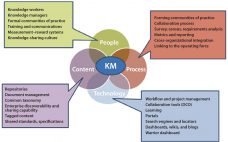
BioPhorum Operations Group Technology Roadmapping, Part 3: Enabling Technologies and Capabilities
Although great strides have been made over the past 20 years to increase the productivity and robustness of manufacturing processes for biopharmaceuticals, the cost and complexity of their development and manufacturing remain high, especially in comparison with those of small-molecule pharmaceuticals. Process improvements are required to increase patient access while maintaining the viability of an R&D-driven biopharmaceutical industry. Facility productivity, cost of goods (CoG), and capital investment all have significant margins for improvement. Such goals can be achieved not only…
Strategies for Successful Sample Transfer
Nadine Ritter is president and senior analytical advisor of Global Biotech Experts, LLC and a long-time member of BioProcess International’s editorial advisory board. At a recent CASSS North American CMC Strategy Forum called “Methods on the Move: Addressing Method Transfer Challenges,” she discussed the biopharmaceutical industry’s logistical challenges of analytical test samples for drug substances and products. At the conference, BPI’s editor in chief Anne Montgomery met with her to discuss some key points of this topic. Logistics Challenges Montgomery:…
How to Set Up a Perfusion Process for Higher Productivity and Quality
Biotherapeutic proteins usually are produced by either fed-batch or perfusion processes. Perfusion manufacturing can provide much higher levels of productivity than fed-batch systems can, thereby reducing production costs. A 2013 study showed that perfusion is more cost effective than fed-batch processes for most combinations of titers and production volumes (1). Moreover, because a perfusion process is much closer to steady state than is a fed-batch process, it often produces a more consistent product — especially for molecules that are sensitive…
Going After a Moving Target: New Production Methods Aid in the Flu Fight
The traditional method of manufacturing vaccines for influenza involves infecting hens’ eggs with the virus, then harvesting and purifying the large amounts of virus that they produce as a result. It’s time-consuming and expensive, requiring large specialized facilities for production. With the advent of genetic engineering and decades of improvement in protein production through cell-line engineering and industrial culture, it was only a matter of time before the vaccine industry saw the real value in modern biomanufacturing instead (1, 2).…
Biosimilar Markets and Regulation: Which Countries Are Going All In?
The pipeline of follow-on (biosimilar and biobetter) products in development for the US, EU, and other major markets is very healthy. It includes nearly 800 biosimilars, about three-quarters of which are presumed to be targeted for major markets, and about 500 biobetters in development. Nearly 1,200 follow-on biopharmaceutical products in the development pipeline are intended to compete with more than 100 currently marketed biopharmaceuticals. This is not just an opportunity in the Western world; biosimilars development is expanding globally. But…
The Clinical Side of Biosimilar Development
Biosimilars have become common on pharmacy shelves in Europe. The first biosimilar product — Sandoz’s Omnitrope version of Lilly’s Humatrope (somatropin) — was approved by the European Medicines Agency (EMA) in 2006. In the decade that followed, more than 20 biosimilars have gained regulatory approval in Europe. The first biosimilar monoclonal antibodies (MAbs) — comparators to Janssen’s Remicade (infliximab) — were approved in 2013. The pace of approvals in the United States has been much slower. The US Food and…
Evolving Bioassay Strategies for Therapeutic Antibodies: Essential Information for Proving Biosimilarity
The modern age of biologics began 35 years ago with the approval of Lilly’s Humulin product — a biosynthetic form of human insulin derived from recombinant DNA and microbial cell culture (1). Today, about a quarter (27%) of the drugs approved yearly by the US Food and Drug Administration (FDA) and European Medicines Agency (EMA) are biopharmaceuticals: primarily monoclonal antibodies (MAbs), but also vaccines, blood products, and (recently), advanced therapies based on genes and cells. A decade ago, the average…
Extractables and Leachables: Standardizing Approaches to Manage the Risk
The implementation, maturation, and benefits of single-use technologies in biopharmaceutical development and manufacturing are well documented and understood. As analytical methods and testing services also rapidly improve, it is clear that management of risk associated with extractables and leachables also must evolve. Standardization is universally accepted as a goal; how to define, implement, and educate the industry is where debate resides. The container–closure segment has had more experience dealing with leachables and extractables than those implementing single-use process components do…