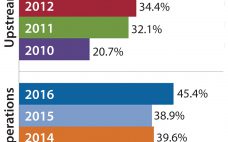
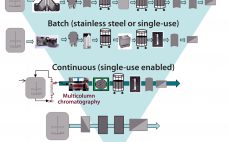
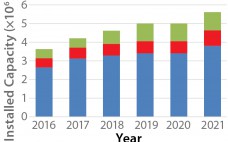
India’s position as a global participant in small-molecule generic drugs, vaccines, and enzymes has been proven over decades. The country is one of the most populous and fastest-growing regions in the world, both economically and technically. But India’s potential as a biologics participant has not been realized. Its competence as a global biologics producer has not yet caught up. Global industry concerns regarding the country’s position in the (bio) pharmaceutical industry haven’t changed much over the past eight years since…
Economics
Funding for Life-Science Ventures: Accelerating Innovation in Tools and Services
As a cofounder of Wave Biotech (now a division of GE Healthcare), my partners and I often struggled with critical choices regarding partnering and funding opportunities. Every new, attractive, and potentially disruptive technology will court attention once it experiences some modest adoption and acceptance, even while attempting to “fly under the radar” of major players. The challenge for life-science entrepreneurs is how best to navigate those decisions and select the right path as company founders. Weighing and evaluating potential partners…
Reducing Clinical-Phase Manufacturing Costs: Collaborating for Savings without Compromising Quality or Performance
In downstream purification of monoclonal antibodies (MAbs), the single greatest contributor to manufacturing costs is the expensive capture step typically based on protein A affinity chromatography. Almost since its introduction to bioprocessing, efforts have been made to reduce the cost of this step. Several alternative ligands have been promulgated as potential replacements for protein A, but they have proven difficult to adopt and scale up. Supplier companies have pushed for increases in capacity and economics, but those are always accompanied…
Outsourcing of Buffer Preparation Activity Is Increasing
The major fluid products used in bioprocessing — culture media and buffers — are classically prepared in-house by rehydrating (dissolving and mixing) powders purchased from suppliers. Most bioprocessing facilities consider in-house preparation of these fluids to be a core bioprocessing task. However, some companies are outsourcing the work either by purchasing preprepared materials from vendors or hiring contract manufacturing organizations (CMOs) to prepare them. Buffer fluid preparation is one area of downstream production operations that are seeing an increase in…
Designing the Optimal Manufacturing Strategy for an Adherent Allogeneic Cell Therapy
Cell therapies (CTs) offer potential treatments for a wide range of medical conditions (1–6) by replacing cells, repairing tissues affected by either disease or damage (7), or delivering genetic or molecular agents that promote self-healing (8). CT research and development is continuously growing (9), with increasing numbers of CT candidates reaching phase 3 clinical trials (9–11). Developers aim to make products that can survive in a competitive landscape while complying with stringent regulatory requirements to control the quality and safety…
Collaboration Is Key to Innovation in Biotechnology
A new report from Thomson Reuters shows that innovation in biotechnology declined slightly in 2015, and biotech is the only one of a dozen worldwide industries examined to show that kind of decline (1). To measure innovation, compilers used metrics such as patents filed and scientific literature cited. Looking at the details, however, the dip was just a 2% drop from 42,584 events in 2014 to 41,624 in 2015. The same dynamics had revealed a 7% increase in innovation from…
Addressing the Challenges of Developing Biopharmaceutical Drugs
The biopharmaceutical industry is enjoying considerable success. Its products account for about a fifth of world pharmaceutical revenues, which are growing at twice the pace of those generated by most traditional chemically synthesized drugs. Biopharmaceuticals populate the list of best-selling drugs, and a number have achieved blockbuster status. Biotechnology stocks have outperformed the general market as investment has flowed into the industry. As with other highly profitable markets, the market for biopharmaceuticals has become increasingly competitive. Reflecting this fact, in…
Managing Customer and Regulatory Expectations
Partnering with a contract development and manufacturing organization (CDMO) allows drug-product sponsors to turn fixed costs into variable costs. Market forecasting by pharmaceutical companies drives numerous decisions in development programs: sales-force resources, geographic resource distribution, and (of course) manufacturing planning. It is a widely accepted fact in the pharmaceutical industry that accurate forecasting is a challenge, especially for new drug launches. A number of models can be used to develop drug forecasts, but none of these models is perfect. No…
Standardized Economic Cost Modeling for Next-Generation MAb Production
Historically, in generating material for clinical testing during antibody process development, emphasis was placed on efficacy, product quality, regulatory compliance, and speed. As the biopharmaceutical industry has matured (and with increasing competition), emphasis has shifted toward cost optimization and manufacturability. Reducing the costs of medicines for patients and payers (thereby broadening access to drugs) is now a key driver during development of new therapies as well as modernizing processes for existing molecules. Cost reduction includes providing robust manufacturing processes that…
From CMO to CDMO: Opportunities for Specializing and Innovation
Biopharmaceutical contract manufacturing organizations (CMOs) were initially enabled when the requirement for a company to file for both an establishment license application and a product licensing application transitioned to the current format of a biologics license application (BLA) submission for biological products (1). The initial focus of such CMOs was to provide large-scale, commercial manufacturing for companies that had already developed and validated bio manufacturing processes. Consequently, CMOs were generally formed as stand-alone service providers that “rented” manufacturing capacity to…