Jim Furey (general manager, PendoTECH) BPI Theater @ INTERPHEX, April 26, 2016 11:30 am–12:00 pm Located in Princeton, NJ, PendoTECH was founded in 2005. The company develops equipment for single-use systems and promotes the concept of automation. It produces sensors, monitors, transmitters, and standard process control systems with the ability to collect and store data, as well as customized solutions. PendoTECH staff have many decades of experience in product development, project management, embedded software development, graphical-interface programs, and mechanical engineering.…
BPI Theater
Significant Technology Advances Enabling Integrated Continuous Bioprocessing
Peter Levison (senior director, Pall Life Sciences ) BPI Theater @ INTERPHEX April 26, 2016, 1:40–2:00 pm Pall’s advancements in integrated continuous bioprocessing include new product launches. The company has built a continuous laboratory at its facility in Westborough, MA. It is 6 m × 6 m and can produce >2 g of pure monoclonal antibody (MAb) per hour. One continuous process flows through one step to the next, from cell culture to formulation. Continuous manufacturing is being used successfully…
Delivering Unique Sterile Primary Container Closures with Advanced Aseptic Manufacturing
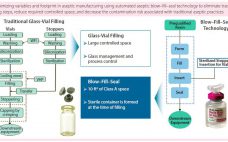
Bill Hartzel (director of strategic execution, Catalent Pharma Solutions) BPI Theater at INTERPHEX, April 27, 2016 11:00–11:20 am How do biopharmaceutical companies leverage technology to provide unique primary container– closures to the market space? Catalent uses an advanced aseptic process of blow–fill–seal (BFS) technology. Adding automation makes for an efficient process that can reduce or eliminate human intervention needed in critical drug-product filling applications. Catalent’s system uses one automated machine in which a container is quickly formed, filled, and sealed…
Case Study: Overcoming the Design Challenges of Expanded Bed Adsorption Columns
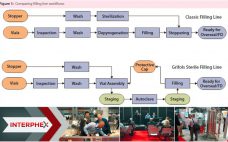
Eduard Noguera (managing director, Grifols) BPI Theater @ INTERPHEX, April 27, 2016, 11:20–11:40 am Hematologist Dr. Jose Antonio Grifols pioneered blood banking and transfusions and founded the Grifols company in Spain, 1940. Present-day Grifols specializes in plasma and protein therapies. Grifols Engineering focuses on design and equipment for process engineering. The speaker has worked for this company for about 15 years in process engineering, including fill-finish processes. He described the company’s invention — the Grifols filling method — which prevents…
Utilizing Environmental Monitoring to Ensure Facility State of Control
James D. Vogel (founder and director, The BioProcess Institute) BPI Theater @ INTERPHEX, April 27, 2016, 11:40 am–12:00 pm James Vogel introduced the concept of trending and how to use it to improve a manufacturing facility’s performance. Trending is defined as collecting data and then examining that information for trends — for example, modeling data to forecast the weather. Sometime a simple graph tells you as much as more advanced statistics can. The key is to pay attention to present…
Getting the Most in Training for New Technologies
A BPI Theater Roundtable at Interphex 2016 On Wednesday, 27 April 2016, BioProcess International and the Biomanufacturing Training and Education Center (BTEC) at North Carolina State University (Raleigh, NC) presented an afternoon training symposium in the BPI Theater at Interphex 2016. Brian Caine, BPI’s cofounder and publisher, offered some opening remarks: “The purpose of these theaters and BioProcess International magazine is to talk about trends and their combined impact on bioprocessing. With the tremendous amount of new technology and products,…
Emerging Biotherapies and Their Manufacturing Challenges
A BPI Theater Roundtable at the 2016 BIO Convention On Tuesday, 7 June 2016, Patricia Seymour (senior consultant in process development at Bio Process Technology Consultants) chaired a lunchtime roundtable titled, “Emerging Bio-Therapies and Their Manufacturing Challenges.” She brought together a panel of three industry experts: Mark Angelino (senior vice president of pharmaceutical sciences at BlueBird Biotechnology) Andreas Weiler (global business unit head of emerging technologies at Lonza) Chris Chen (chief executive officer of WuXi Biologics). Mark Angelino (BlueBird Biotechnology)…
Industrialization and Commercialization of Gene and Cell Therapies
A BPI Theater Roundtable at the 2016 BIO Convention On Tuesday, 7 June 2016, Mike Ward (global director of content at Informa) chaired an afternoon roundtable titled, “Challenges Associated with the Industrialization and Commercialization of Gene and Cell Therapies.” He brought together a panel of four experts: Morrie Ruffin (managing director, Alliance for Regenerative Medicine, ARM) Michael Werner (ARM’s executive director) Sarah Haecker Meeks (chief scientific officer, Adjuvant Partners; director of technology sections, ARM) Tom Novak (vice president of strategic…
How to Manufacture and Deliver the Drugs of Tomorrow
Andreas Weiler (global business unit head for emerging technologies, Lonza), BPI Theater @ BIO, June 7, 2016, 11:00–11:20 am How do we know what the future will look like? If we look at preclinical products in the drug development pipeline, we know what is likely to come to the market over the next 5–10 years. We don’t know exactly which products will succeed, but we know the kinds of products that will be available. And a company such as Lonza…
Recent Advances for Rapid Development of High-Quality, Robust Mammalian Cell Culture
Jozef Orpiszewski (senior director of program design, Fujifilm Diosynth Biotechnology), BPI Theater @ BIO, June 7, 2016, 11:20–11:40 am Fujifilm Diosynth Biotechnology is focused on accelerating bioprocesses from gene to finished products by shortening the time line, all while following quality by design (QbD) precepts and making processes as robust as possible. When this CMO started several years ago, the development time line was about 18–24 months. Using new tools, approaches, and innovations, the company has shortened that time to…