A BPI Theater Roundtable at Interphex 2016 On Tuesday, 26 April 2016, Eric S. Langer (managing partner of BioPlan Associates) chaired a morning roundtable titled “Deciding on Single-Use vs. Stainless Steel Strategy: What the CMOs Know That Biopharma Needs” as a follow-up to a similar discussion held last year. Langer brought together a panel of four contract manufacturing organization (CMO) industry experts: Daniel Vellom (senior director of global technology innovation at Sanofi Pasteur) Sue Behrens (senior director of process technology…
Author Archives: Bill Hartzel
Delivering Unique Sterile Primary Container Closures with Advanced Aseptic Manufacturing
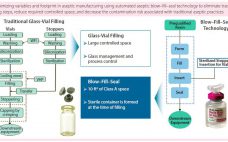
Bill Hartzel (director of strategic execution, Catalent Pharma Solutions) BPI Theater at INTERPHEX, April 27, 2016 11:00–11:20 am How do biopharmaceutical companies leverage technology to provide unique primary container– closures to the market space? Catalent uses an advanced aseptic process of blow–fill–seal (BFS) technology. Adding automation makes for an efficient process that can reduce or eliminate human intervention needed in critical drug-product filling applications. Catalent’s system uses one automated machine in which a container is quickly formed, filled, and sealed…
Manufacturing Strategies: BPI Theater @ Interphex 2015
Bill Hartzel (director of strategic execution for advanced delivery technologies, Catalent Pharma) 1:30–1:55 pm Reducing the Risk Associated with the Filling of Biologics with Advanced Aseptic Processing Hartzel discussed leveraging “blow–fill–seal” (BFS) aseptic processing technology to reduce risks associated with biologics fill and finish. A number of drug recalls have been associated with microbial and particulate contamination of glass vials, problems that could be solved using BFS. Automation eliminates human intervention at this critical stage, driving out associated risks. Hartzel…