As part of BPI’s “Ask the Expert” series, editorial advisor William G. Whitford (senior technical market manager for GE Healthcare Life Sciences) spoke with editor-in-chief Anne Montgomery and marketing and digital content strategist Leah Rosin on two separate occasions about issues related to culture media and expression titers. Sourcing Serum-Free Media Anne discussed cell culture media and process fluids with Bill in March 2014. Whitford: Things are advancing, and the industry is changing significantly. In general, we are using more…
2015
QBD Manufacturing Strategies: The CMC Strategy Forum Series, Part 2
The CMC Strategy Forums provide a venue for biopharmaceutical product discussion. They focus on relevant chemistry, manufacturing, and controls (CMC) issues throughout the life cycle of a therapeutic and thereby foster collaborative technical and regulatory interaction. Forum chairs share information with regulatory agencies to help them merge good scientific and regulatory practices. Outcomes of the forum meetings are published in BioProcess International and on the CASSS website. This process is meant to help ensure that biopharmaceutical products manufactured with advancing…
New Paradigms for Process Validation: A Practical Approach
Both the United States and the European Union offer guidance on a life-cycle approach to process validation. This goes beyond the traditional three to five lots run at the center point of proposed ranges for operating parameters. New approaches leverage product design and process development information. They facilitate adapting the QbD paradigm to allow for a science- and risk-based selection of critical process parameters, key process indicators, and appropriate specification criteria. The number of runs for process performance qualification (PPQ)…
Raw Material Control Strategies for Bioprocesses
The 15th WCBP CMC Strategy Forum, “Raw Material Control Strategies for Bioprocesses,” met on Sunday, 11 January 2009 in San Francisco, CA. This forum considered the design and implementation of control strategies for complex raw materials used in bioprocessing. Discussion focused on key approaches and application of risk assessment tools that can be used to identify and assist in mitigating potential safety and efficacy concerns that can affect the quality of biological products. Two Sessions To fully explore the topic,…
Multiproduct Facility Design and Control for Biologics: Challenges and Considerations
Multiproduct facilities are increasingly integral to corporate biologics network and supply chain strategies. Manufacturing capacity strategies ensuring appropriate facility design and procedural controls to manage the risks of producing multiple products are critical to the successful deployment of commercial and clinical supply plans. A Chemistry, Manufacturing, and Controls (CMC) Strategy forum was held in Bethesda, MD, in August 2011 to highlight various challenges, risks, and control strategies associated with multiproduct facilities. Multiproduct strategies for the manufacture of a variety of…
Drug Products for Biological Medicines: Novel Delivery Devices, Challenging Formulations, and Combination Products
The importance of investing science and technology into drug product development has become evident as different product types, higher protein concentrations, and doses and requirements for improved delivery of biological drug products have increased. The need to give patients larger and more concentrated doses has challenged formulation scientists and driven development of new technologies that can deliver those doses. Delivery devices fall under device regulations and have distinctly different design, development, and validation requirements from those of protein drug products…
BioProcess International European Summit 2015 – Speaker Interviews
Listen to exclusive interviews with speakers at the BioProcess International European Summit 2015. Find out more about the program in the March 2015 Special Report.
Toward Industry Standardization of Extractables Testing for Single-Use Systems: A Collective BPSA Perspective
Here we present a consensus position of the membership of the Bio-Process Systems Alliance (BPSA), the trade organization for the single-use industry based in Washington, DC. BPSA’s membership includes 48 corporate and institutional entities, among them component suppliers, systems integrators, end users, and independent testing laboratories. Consensus within this membership is reached through an official ballot of representative voting members, as provided for in the organization’s by-laws. The position outlined below was approved by such an internal consensus-balloting process. Building…
Extractables Profiles: A Comprehensive Approach Produces Long-Term Results
Biopharmaceutical manufacturers spend years developing and testing new drug and biologic products to ensure their efficacy, safety, and usability for patients. Such knowledge is extremely valuable, but those same principles often get overlooked in selection of packaging and delivery systems. Decisions on packaging and delivery often are made almost as an afterthought. However, issues related to components such as extractables and leachables can affect patient safety and product quality. In addition, a lack of extractables and leachables data in filings…
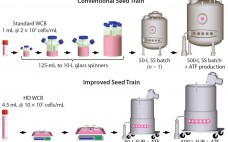
A Novel Seed-Train Process: Using High-Density Cell Banking, a Disposable Bioreactor, and Perfusion Technologies
A typical cell culture process begins with thawing of a cryopreserved cell-bank vial, followed by successive expansions into larger culture vessels such as shake flasks, spinners, Wave bags, and stirred bioreactors (1). When culture volume and cell density meet predetermined criteria, the culture is transferred to a production bioreactor in which cells continue to grow and express product. This approach presents several challenges. Shake flasks or spinners used in the initial stages require manual manipulations inside a laminar flow hood,…