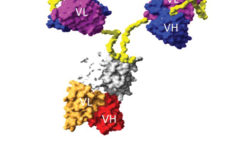
This webcast features: James Brady, PhD, VP of Technical Applications and Customer Support, MaxCyte, Inc. Moving bispecific and complex protein therapeutics from the bench to the clinic requires a scalable process that is consistent and reproducible while delivering a biologically active product. MaxCyte’s delivery platform is a high performance, electroporation-based technology that can rapidly generate milligram to gram quantities of protein in the cell line of choice. You will learn: How to express quality bi-specific antibodies, bi-specific T cell engaging…
Upstream
Synergizing Transient and Stable Protein Expression for Accelerated Biotherapeutic Development
This webcast features: Krista Steger, PhD, Scientist, MaxCyte Transitioning early stage discovery efforts with later stage development activities is critical for moving biological therapies into the clinic in an efficient and timely manner. MaxCyte’s scalable Flow Electroporation™ Technology allows gram-scale transient expression of antibodies and other proteins in cell types that are relevant to biomanufacturing. At the same time, the high levels of efficiency and viability provided by MaxCyte transfection, can shorten timelines and reduce labor involved in generating clonal cell…
Challenges of Scale Up and Development of Insect Cell Culture and BEVS
This webcast features: Sharyn Farnsworth, Associate Principal Scientist, FUJIFILM Diosynth Biotechnologies A strategy for scale up and implementation of Insect Cell Culture (ICC) should be assessed at the earliest stages of process development as there are a different set of challenges in the culture of ICC and BEVS versus a CHO culture and a limited number of vendors experienced enough to guide you through the adventure of manufacturing ICC. Each system has a unique problem set ranging from which cell…
Single-use XCell ATF Systems for Continuous Processing: 100% Cell Retention, 80% Faster Set-Up
This webcast features: Christine Gebski, Vice President, Product Management and Field Applications, Repligen In today’s flexible manufacturing environments and to improve manufacturing cost of goods, many companies have implemented single-use equipment. To meet these changing customer requirements, the XCell™ ATF 2 and ATF 6 are now available in single-use formats. The XCell ATF Single-use systems deliver the same cell culture performance as the stainless steel systems but with 8x faster set-up time and a simplified autoclave-free implementation workflow. These elements…
Taking Medium and Feed Development Beyond Maximizing Protein Titer to Optimizing Glycan Distribution and Simplifying Process Scale-Up
This webcast features: Serena Fries Smith, Process Science Manager, Thermo Fisher Scientific In the early 2000s when many processes were struggling to achieve 1 g/L, maximizing titers was the industry’s biggest challenge and was essential to having favorable cost of goods and an economically viable product. Over the last 1–15 years, the industry has made significant advances in media and feeds. Due to these advancements, today a standard fed-batch process can typically achieve 3 g/L and some processes are achieving…
Improving Single-Use Bioreactor Design and Process Development: New Research Towards Intensifying Seed-Train and Scale-Up Methods Using 5:1 Turn-Down
This webcast features: Surendra Balekai, Senior Global Product Manager, Thermo Fisher Scientific Bioproduction Operating bioreactor vessels at low working volumes (low turn-down ratio) is often desirable but brings about challenges in regard to mixing, mass transfer, and process control. Research done toward optimizing cell culture has provided methods to improve performance and control when operating under these conditions. Implementing bottom heat exchange, making changes to impeller angle and height, taking advantage of the unique Thermo Fisher drilled-hole sparge design, and…
How to Boost Profits with Single-Use Powder Transfer
This webcast features: Chris Rombach, Director of Global Strategic Accounts, ILC Dover Biopharmaceutical manufacturers face a number of challenges. Open-suite biopharma manufacturing is gaining popularity. But it also leaves surfaces open to contamination, workers at risk for exposure to airborne particulates, and processes more exposed to product loss due to waste or spillage. Liquid handling systems adapted for dry powder applications are often a compromise, because liquid and powder flow rates, handling characteristics and dispensing volumes differ dramatically. A purpose-built,…
Modification of Glycans in Bioprocessing
This webcast features: Ryan Boniface, R&D Scientist, Thermo Fisher Scientific Protein quality is a topic of increasing importance as quality elements can have a significant impact on clinical behavior of a molecule. This talk will explore approaches to modify glycan patterns contribute to predicting, achieving and maintaining preferred glycosylation profiles. Glycosylation is a key product quality attribute for many biotherapeutic proteins expressed in CHO cells. N-linked glycans may display macro- and micro-heterogeneity; the degree of this variation can depend on…
Objective Characterization Data on Drug and Gene Delivery Platforms
This webcast features: Dr. Josefina Nilsson, Head of Vironova Services, as well as her colleagues, Gustaf Kylberg and Mathieu Colomb-Delsuc. Advanced analytics that provide objective and reliable data can help to improve processes and shorten development time of drug and gene delivery platforms. The use of electron microscopy (EM) imaging combined with analysis using the proprietary Vironova Analyzing Software (VAS) enables semi-automated particle detection and classification. Statistically significant results are obtained in a time and cost effective manner. Typical data…
High Performance Cell Retention Technology for Cell Culture Process Intensification
This webcast features: Millie Ullah, PhD, Associate Director of Upstream Product Management and Marketing, Repligen The move toward continuous processing and intensified processes is a paradigm shift in bioproduction. In upstream cell culture, the challenge is to simplify and minimize process steps (N-1 perfusion) while intensifying cell retention and viability (high density cell expansion). This webcast will present a case study where use of the ATF cell retention system in process intensification of a fed-batch culture resulted in successful N-1…