Biosimilars are a relatively new subset of biopharmaceuticals, with the biotechnology industry finally maturing such that off-patent generic-type products increasingly will be entering major markets (1–3). So far, more than 20 biosimilars for a limited number of reference products have been approved in major markets, primarily the European Union. Only two products have been formally approved as biosimilars in the United States. For this rapidly growing industry sector, little consensus or authoritative information is available yet regarding how and where…
Economics
Achieving Competitive Advantage in the Biopharmaceutical Industry
Drug development is a complex process that is associated with high drug-candidate attrition rates, long development times, and high costs (1, 2). Drug development costs have increased over the past two decades, with current average development cost of about US$2.6 billion, of which $1.4 billion is the direct cost (1, 2). On average, drug development takes at least 10 years to market authorization (2). Biopharmaceutical companies often follow a strategy of developing drugs for multiple clinical indications and biological targets…
Opportunities in Latin America Beyond the Olympics
Olympics fans aren’t the only ones turning their eyes toward Brazil. According to JLL’s fourth annual Life Sciences Outlook, the 2016 Summer Games host nation is also one of the world’s top 10 “Global Clusters to Watch,” thanks to its proven capacity for medical device manufacturing and a growing healthcare market. Increasingly, Latin America overall has been on the radar for life sciences companies seeking greater operational efficiency and opportunity, expanded market access, promising demographics, lower-cost land and labor, and…
Developing Markets Bring Opportunity But Not Without Real Estate Risk
Life-sciences markets develop in their own way, but one aspect they all have in common is the unpredictability of their growth. Not only are local economies unpredictable, but so are companies and their needs. Driven by competing pressures to seek new markets and new innovations while operating more efficiently, biopharmaceutical companies are increasingly setting their sights on new horizons abroad. Those investments are not without risks, however. Real estate is at the core of overseas expansion, and that’s where it…
Hiring and Staffing in Biopharmaceutical Manufacturing: Five-Year Trends Indicate Difficulty in Filling Positions
The biopharmaceutical manufacturing industry continues to expand rapidly, with growth in revenue for both suppliers and drug innovators averaging about 15% per year. Understandably, much of the industry’s recent focus has been on the pace of technological improvements that have boosted productivity and performance. But another issue is becoming just as critical: The flow of skilled staff able to keep up with the industry’s growth isn’t keeping pace. Results from our 2015 annual industry study suggest that the hiring market…
Bioindustry Researchers Take Strides in Preventing Diseases: Developments in Genetic Diagnosis and Therapy
Johnson & Johnson’s pharmaceutical research arm recently launched a drug-development project that will intercept diseases before patients even realize they’re sick. In the company’s words: “The future of healthcare will increasingly depend on identifying and correctly interpreting the earliest signals of disease susceptibility, preventing or intercepting disease before it even begins.” (1). The first targets include Alzheimer’s, some forms of cancer, heart disease, rheumatoid arthritis, multiple sclerosis, and type 1 diabetes. The company’s Disease Interception Accelerator group will search for…
Accounting for Portfolio Risk: Creating an Analytical Framework for Optimizing the Portfolio Mix
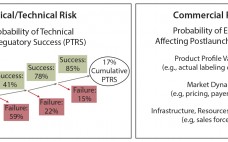
In the biopharmaceutical industry, portfolio risk is categorized as clinical/technical or commercial (Figure 1). Both types pose challenges to quantify. Portfolio managers tasked with optimizing the mix of products in a company’s pipeline often struggle to create apples-to-apples frameworks that can consistently compare risk across asset categories. The framework described herein can help you account for both technical and commercial risk using simple analytical tools. The foundational analytical element of portfolio management is traditionally the asset forecast: a composite best…
From Silos to Synergy: Optimize Your Managed-Markets Division with Cross-Team Collaboration
Like many other industries, life sciences faces rapid, technology-driven changes and a shifting business environment. Many patients are covered by Medicare, Medicaid, or national healthcare organizations outside the United States. That necessitates pricing and reimbursement negotiations that are complicated by rules and regulations. Patients whose health plans belong under a managed-care organization (MCO) or pharmacy-benefit manager (PBM) have access to prescriptions primarily through staff-model pharmacies, mail-order pharmacies, and network pharmacies. Drug manufacturers offer incentives to such organizations in the form…
Standardization of Disposables Design: The Path Forward for a Potential Game Changer
Recent articles have described how the debate on standardization is slowing down adoption of single-use technology (1). The Standardized Disposables Design (SDD) initiative is working to design simple standard single-use solutions for real-life examples (e.g., buffer bags). In reality, a buffer is a buffer whether it is made in Europe, Asia, or America, so in essence different solutions are not necessary for different end users. A buffer bag is not difficult to design, and it does not vary greatly in…
Effective Management of Contract Organizations: Keeping the Product Pipeline Moving, Compliant, and Available
Both small and large biopharmaceutical companies are increasingly pursuing the outsourcing of manufacturing and testing throughout the product lifecycle. The growing use of contract manufacturing organizations (CMOs) and contract testing organizations (CTOs) has led to increasing complexity within the biopharmaceutical industry as more third-party sites are leveraged to support global markets. To address those issues, a CASSS Chemistry, Manufacturing, and Controls (CMC) Strategy Forum was held in Washington, DC, 27–28 July 2014. The title was “Effective Management of Contract Organizations: Sponsors, Contract Organizations, Health Authorities and Patients — Keeping the Product Pipeline Moving, Compliant, and Available.” The CMC Strategy Forum is…