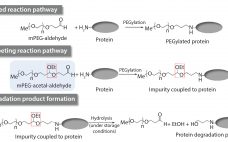
Impurities related to raw materials used for bioproduction can be inadvertently introduced into a manufacturing process, causing potential failure to meet in-process controls or release specifications. Unexpected impurities also can reduce yield and affect the quality, safety, and effectiveness of a final product (1). Raw-material impurities can originate from starting components or reagents used in manufacture. They can be generated in situ during synthesis or as degradation products. Impurities also can result from improper handling, packaging, and storage. Identification and…
Sunday November 16, 2025