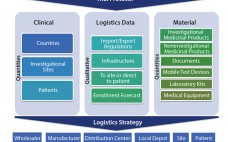
A clinical supply chain fulfills perfectly all four characteristics of what Packowski describes as a “VUCA” (volatility, uncertainty, complexity, and ambiguity) world (1). In commercial markets, supply chains depend predominantly on consumer orders. For global drug development programs, both investigators and patients can be considered end consumers. The international journey of a specific investigational medicinal product (IMP) includes all of the following: global sourcing of comparators, manufacturing, storage, distribution, site/patient (consumer) management, and return and destruction of the IMP. Application…
Author Archives: Jens Mattuschka
A Critical Mission: Clinical Trial Material Storage and Distribution
As if manufacturing of investigational medicinal products (IMPs) weren’t challenging enough already, the appropriate storage and distribution of sensitive biological products can be an adventurous journey itself — especially if not carefully planned and managed. No one solution fits all situations. Many things must be evaluated during planning stages. For example, what is more important: time to delivery or quality of transport and product integrity until drug can be administered to a patient? It is important to understand key storage…