The potential therapeutic value of many proteins — including enzymes, receptors, cytokines, blood factors and peptides — can be realized by fusing them to the Fc region of human immunoglobulin G. Of the 46 monoclonal antibody (MAb) and MAb-derivative products approved by the FDA to date as human therapeutics, 10 are Fc-fusion proteins (Table 2). Among approved products, several structural variations are represented (Figure 4). In BPI’s October 2014 issue, Part 1 of this review examined the structure and manufacturing…
Manufacturing
Sterilization Effects on Elastomer Characteristics and Functionality in Parenteral Delivery Systems
To drive efficiencies in producing parenteral drug products, manufacturers are using containers and closure components that are received sterile and ready to be introduced into filling lines. The effects of sterilization on the properties of ready-to-use (RU) components must be assessed to ensure proper processing techniques and suitability over the components’ intended shelf lives. Sterile-drug manufacturers must determine the best sterilization method for components based on their respective drug products and processes. Critical areas of risk include potential changes related…
Creating Value Through Investment
During my MBA course, Professor Pierre Casse — then at the International Institute for Management Development (IMD) in Lausanne, Switzerland — regularly reminded us that one key to success was constantly finding new ways to “delight and inspire your clients” by creating value. SAFC achieved that objective in its “Overcoming Supply Chain Vulnerability and Lowering Risk in Biopharmaceutical Manufacturing” symposium 17–18th June 2014 in Turnberry, Scotland. Along with a day of industry insight, the event included a visit and tour…
Trends in Setting Single-Use Technology Standards
The biopharmaceutical industry now incorporates single-use (SU) technology and systems in most production processes based on cell culture (1, 2). Implementation of such technologies has led to the availability of prepackaged and sterilized systems complete and ready for use with preinstalled mixers and monitoring probes. From upstream process- material preparation through final-product formulation, biopharmaceutical sponsors are increasingly presented with numerous SU solutions that support all major production platforms (3–5). The number of SU materials and suppliers in biopharmaceutical manufacturing has…
Immunoglobulin Fc-Fusion Proteins Part 1: Their Design and Manufacture
Over the past three decades, 45 monoclonal antibody (MAbs) and MAb-derivative products have been approved for therapeutic use in the United States (Table 1). One class of antibody derivatives is growing in importance: Fc-fusion proteins. Many biologically active proteins, including receptor ECDs (see “Abbreviations” box), cytokines, enzymes, and bioactive peptides have very short serum half lives because rapid renal clearance limits their exposure in target tissue (and, consequently, their pharmacological effect). The primary reason for fusing a biologically active protein…
Replacing Reverse-Phase Chromatography for Mass Spectrometry: Is Salt-Free Size-Exclusion Chromatography Ready?
Protein mass is often determined using ultraperformance liquid chromatography (UPLC) coupled with electrospray-ionization mass spectrometry (UPLC/ ESI MS or simply LC-MS). A UPLC system equipped with an ultraviolet (UV) detector serves as an assisting vehicle to deliver purified and separated protein molecules to the mass analyzer. Reserved-phase chromatography (RPC) is the most common chemistry chosen to serve this purpose. For sample purification, not only does RP-UPLC use salt-free mobile phases that are amenable to MS, but it also can efficiently…
Banking Cord Blood for Stem Cell Research
Cord blood is becoming an increasingly popular and important topic of discussion among expectant parents. It comes from a newborn’s umbilical cord and contains hematopoietic stem cells (HSCs), which are the building blocks of a body’s blood and immune system. After a baby is born, cord blood is routinely discarded as medical waste — unless the parents choose to have what blood remains in the umbilical cord collected. Presently, more than 90% of cord blood is discarded, limiting the potential…
Antibody Drug Conjugates: The State of the Art
Monoclonal antibodies have dominated the biopharmaceutical market for over two decades. Few people doubt that their future success in fields such as oncology, inflammation, and autoimmune diseases owes much to the development of antibodies conjugated to cytotoxic drugs. Like all great innovations, this is a breathtakingly simple concept: Combine the targeting specificity of an antibody with a small-molecule drug as an effector component joined to that antibody by means of a small chemical or peptide linker, and you have a…
Bioprocessing Challenges of Antibody–Drug Conjugates
Development of highly potent active pharmaceutical ingredients (HPAPIs) is clearly a pharmaceutical industry trend. Highly potent drug products involve active agents and APIs that are so potent therapeutically (or simply just outright toxic) even in small dose that special precautions are required during their manufacture — particularly when handling the active agents. Such requirements include maximal containment and isolation of the process stream. Worker exposure and environmental release clearly pose problems. The necessity and intensity of containment efforts with HPAPIs…
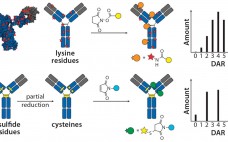
The Next Step in Homogenous Bioconjugate Development: Optimizing Payload Placement and Conjugate Composition
[Audio Recording] Bringing a new biologic drug to market is a long and expensive process, with research and development (R&D) cycles that can span up to 15 years and may cost over a billion dollars. Biologic drug development also involves significantly more complex manufacturing and CMC components than does development of small molecules. Nonetheless, the pharmaceutical industry is increasingly shifting its R&D efforts to focus on biologic drugs. According to a recent report from Tufts Center for Study of Drug…