The increasing penetration of disposable devices in the biopharmaceutical manufacturing industry has been well documented, and with good reason. These applications represent a new paradigm in the evolution of biomanufacturing technologies and practices, opening the industry up to new possibilities such as flexible and modular facilities. But stainless steel applications and innovations remain vital to this industry. Some stainless devices may be irreplaceable, including large tanks, autoclavable and heat-sterilized fermentors and bioreactors, and storage and filling devices. Other innovations in…
Manufacturing
Disposable Components in Biomanufacturing: A Regulatory Perspective
On a holistic level, the decision regarding whether to use single-use or stainless steel equipment in a biomanufacturing plant is a significant one. It greatly influences the design, construction, layout, and operation of a plant — and consequently, the timing and cost of the overall project. And regulatory review can add an element of risk to using anything new. Disposables have been viewed as a revolutionary concept, and they are. But from a routine regulatory operational perspective, I believe it…
The Single-Use or Stainless Steel Decision Process: A CDMO Perspective
Decisions regarding whether and when to use single-use (SU) (disposable) devices or stainless steel (SS) equipment for biopharmaceutical manufacturing have been discussed for more than a decade. To date, no argument in terms of safety, cost-effectiveness, or operational efficiency is fully convincing to choose one technology platform or the other for all applications. Biopharmaceutical companies often do not have in-use data to make strategic manufacturing decisions. But one group has been significantly growing its expertise in use of single-use technologies:…
Meeting the Demand for a New Generation of Flexible and Agile Manufacturing Facilities: An Engineering Challenge
Classical stainless steel installations in purpose-built facilities dominate the global capacity for commercial biopharmaceutical manufacturing. Early facilities that were designed for single-product processes are now aging, putting them on the investment radar for upgrades to enable manufacturing diversity, and allow more efficient facility use. More than ever before, global engineering leaders are confronted with complex strategic and financial decisions when they seek to invest capital in new flexible pharmaceutical facilities or flex-grading aging facilities for supply of pipeline products. Manufacturing…
End-User Considerations: How Biopharmaceutical Companies and Their Employees Approach Single-Use and Stainless Steel
Along with the other perspectives offered in this special issue, people tasked with actually implementing and working with single-use, stainless-steel, or the more common variations of hybrid systems deserve a say also. When seeking out end-user comments, however, we should consider both the overall company decisions about which systems to use (decisions that may not have accommodated much input from the manufacturing floor) and opinions from individual scientists, technicians, and process engineers themselves who are presented with the facility design and…
Single-Use and Stainless Steel Technologies: Comparison, Contrast — Where Are They Going?
At a recent conference, there was much talk about a major biopharmaceutical project in Asia for which the owner of a company chose a design with primarily traditional multiuse stainless steel tanks and processing equipment, rather than single-use polymer equipment. There were even some stainless steel equipment vendors toasting at the bar that this may be the beginning of a “renaissance” of stainless steel. Is the single-use systems (SUS) honeymoon over? Are many industry professionals hoping this is the end…
Standardization of Disposables Design: The Path Forward for a Potential Game Changer
Recent articles have described how the debate on standardization is slowing down adoption of single-use technology (1). The Standardized Disposables Design (SDD) initiative is working to design simple standard single-use solutions for real-life examples (e.g., buffer bags). In reality, a buffer is a buffer whether it is made in Europe, Asia, or America, so in essence different solutions are not necessary for different end users. A buffer bag is not difficult to design, and it does not vary greatly in…
From Chips to CHO Cells: IT Advances in Upstream Bioprocessing
Advances in our capabilities for data acquisition, storage, and manipulation are providing the biopharmaceutical industry with an increased understanding of what must be controlled in bioproduction as well as the ability to control it. Developments in hardware, processing algorithms, and software are changing the landscape of bioprocess administration. Increased power for information gathering and processing began with the remarkable increases in microprocessor speed, pipelining, and parallelism over the past couple of decades (1); it continues with advances in data handling…
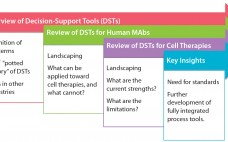
Decision-Support Tools for Monoclonal Antibody and Cell Therapy Bioprocessing: Current Landscape and Development Opportunities
Industrial-scale manufacturers in a number of fields — from automobiles to biotherapeutics — have long relied on powerful computational and mathematical tools to aid in the scale-up, optimization, quality control, and monitoring of product development (1–5). Typical process pathways are highly multifactorial, with numerous branch points, feedback steps, instrumental attributes, and target parameters. Moreover, margins for error are minimal for most industrial processes, requiring high standards of precision from industrial and operational pathways (6). For those reasons, the complexity of…
Primary and Higher-Order Structural Characterization Strategy for Biosimilarity Assessment
The development pathway of a biosimilar is unlike that of a novel biotherapeutic. Many regulatory authorities reference a stepwise approach to establishing biosimilarity. Analytical requirements are greatly increased before a product enters clinical testing. Enhanced analytical efforts entail physical, chemical, and biological characterization of a biosimilar product compared with an originator reference product. Strategies at this early stage must include intensive characterization of multiple batches to determine variability of quality attributes. Here I address some issues involved in meeting regulatory…