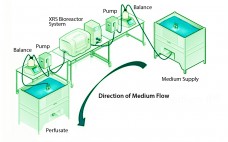
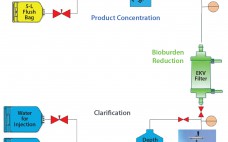
Although implementation of continuous manufacturing for biopharmaceuticals is in the early stages, continuous cell culture technology has been around for close to thirty years. Perfusion was initially developed in the late 1980s as a means for increasing protein titers (1). However, high costs driven by media consumption limited widespread commercial adoption. In the same time frame, advances in cell line engineering, media composition, and bioreactor design led to 10-fold increases in titers for batch and fed-batch modes, eliminating the first…
Manufacturing
Flexible Automation for Continuous Unit Operations
Continuous processing has the potential to provide significant cost and time savings for biopharmaceutical manufacturing, but that potential can be realized only if appropriate automation solutions are available for continuous flow between disparate upstream and downstream operations. Pall Life Sciences’ Allegro MVP system, a fully automated bioprocessing system designed for use in upstream and downstream single-use processing, enables flexible automation and thus facilitates continuous biopharmaceutical manufacturing. This article presents the results obtained using the Allegro MVP system in combination with…
Bacteriophages, an Alternative to Antibiotics: Challenges and Possible Solutions for Bringing Them to Market
Bacteriophages are viruses (consisting of a genome contained within a protein coat) that specifically infect bacteria. They are the most abundant living entities on earth — the estimates range from 1030 to 1032 in total — and play key roles in regulating the microbial balance in every ecosystem where that has been explored (1). Bacteriophages are genotypic and phenotypically different from viruses that infect Archaea (Archaeovirus) and Eukarya (Eukaryovirus). The name bacteriovirus has been proposed as scientifically more accurate (2).…
Bridging Polymer Science and Biotechnology Applications with Single-Use Technologies
Implementation of single-use technology in the biotechnology industry is increasing every year. One major interest has been understanding the interaction of extractables with protein and cells for applications ranging from cell banking to biopharmaceutical manufacturing. In October 2015, the Engineering Conference International (ECI) organization hosted a conference in Leesburg, VA, to explore how the science of plastic applies to bioprocessing. The “Single-Use Technologies: Bridging Polymer Science to Biotechnology Applications” meeting brought together experts from different fields to share issues, understanding,…
Osmolality Measurements for High-Concentration Protein–Polymer Solutions: Variation Based on Working Principles of Osmometers
Osmolality is a critical attribute for injectable formulations. It is desirable to have products match physiological osmotic conditions. Furthermore, osmolality provides confirmation of soluble content in solution. Preventing injection of hypo- or hyperosmotic solutions is a key element of parenteral formulation development. Additionally, some investigators have explored correlations between injection pain and formulation osmolality, although no significant correlation has yet been observed (1–4). Osmolality is a valuable in-process test also because it provides a reliable and repeatable value that reflects…
Enhanced Biosimilar Product Characterization: A Case Study Using Raman Spectroscopy Combined with Dynamic Light Scattering
Biophysical characterization has drawn great attention from the biopharmaceutical industry and regulatory agencies across the globe, especially for use in biosimilar drug product development. Currently available biophysical characterization tools can help in screening and optimizing better (more stable) formulations for such products. However, most tools cannot be used for head-to-head comparison of the biophysical properties of an optimized biosimilar formulation with those of an innovator product at higher concentrations. We developed and optimized a formulation for monoclonal antibody MAb B…
A Single-Use, Clinical-Scale Filling System: From Design to Delivery
Single-use components have been successfully incorporated into many unit operations for both upstream and downstream processing, from laboratory scale to commercial manufacturing. The development of single-use filling needles has created an opportunity to introduce fully disposable systems into final formulation and filling of drug products (1). One major challenge in replacing a cleanable filling line containing stainless steel needles is to ensure that an alternative system can satisfy all critical performance parameters established for an existing process. In 2012, Merck…
Single-Use Technology Enables Flexible Factories
The biosimilars market is rapidly evolving, with more than 450 biosimilar molecules in development worldwide, and many anticipated transfers of molecules in process around the globe. With US$85 billion of biopharmaceutical products coming off patent by 2020 (1), the driving force to develop biosimilars is strong. The market will be highly saturated, with dozens of biosimilars currently in development for each current blockbuster molecule. We know of 46 trastuzumab biosimilars and 39 rituximab biosimilars in development. Because the biosimilars market…
Deciding on an Integrated Continuous Processing Approach: A Conference Report
Biopharmaceutical manufacturers are increasingly investigating or implementing continuous processes, and industry experts are participating in vibrant and healthy discussions on a wide range of process approaches being adopted. We seem to have entered a phase within the biopharmaceutical industry (which is often described as conservative) of intense bioprocess innovation as manufacturers strive to lower costs and provide agile, responsive supply chains for “drug on demand” continuous manufacturing operations. Senior industry leaders met in Berkeley, CA, in late 2015 at ECI’s…
May Supplement Introduction: The Evolving Outsourcing Culture
When I began recruiting contract manufacturing organizations (CMOs) and contract development and manufacturing organizations (CDMOs) to join the Pharma & Biopharma Outsourcing Association two years ago, some of the biologics-focused companies were concerned about being in a trade association alongside small-molecule–focused providers. It wasn’t quite “Upstairs, Downstairs,” much less a Sharks vs. Jets rivalry, but they weren’t sure whether my big-tent approach was viable. (For you millennials in the audience, please replace those references with “Downton Abbey” and “Batman v.…