For an industry built on constant change, there’s a surprising disconnect between the continuous drive for innovation and the inflexible facilities that house biopharmaceutical operations. Some of today’s facilities are built for today’s use with little thought about tomorrow’s. The typical approach for a new process or drug coming to market is to start with a brand-new building and permanently embedded equipment designed around that specific process. That approach is expensive and unsustainable. New bioproduction facilities can cost US$500 million…
Manufacturing
Conference Report: The Drug Product Track at 2017’s BioProcess International Conference and Exhibition in Boston, MA
At the Hynes Convention Center in Boston, MA, during Knect365’s “Biotech Week Boston” in late September of 2017, one track of the BioProcess International Conference focused on drug products, fill–finish, and formulations. Presenters represented a number of major biopharmaceutical companies — AbbVie, Amgen, Biogen, Eli Lilly, Genentech (Roche), GlaxoSmithKline, Johnson & Johnson, Lonza, Pfizer, and Sanofi — as well as suppliers Bosch, Merck (MilliporeSigma), ReForm, and Single-Use Support. They focused on predictive modeling, quality by design (QbD) and process analytics,…
A Vaccine Case Study: Qualifying Redundant Disconnection Technologies As Container-Closure Systems for Long-Term Storage and Shipping
The expanding complexity of biopharmaceutical manufacturing puts increasing pressure on single-use systems to meet the demands of the modern industry’s global footprint. Individual sites within a given organization often are specialized to a fixed number of “modular” process steps (1). Such product segregation increases plant efficiency and output while making the best of staff competencies. But it also can create an additional need for transportation of intermediate or bulk drug substance (BDS) over long distances. Freezing generally is used to…
Beyond Compliance: Serialization Has More to Offer
As the global pharmaceutical industry implements serialization (track and trace from manufacturing to dispensing) to meet governmental regulatory requirements, other opportunities arise for drug companies. The main driver here is to improve the integrity of the overall drug supply chain, but other meaningful business benefits can come from serialization. Generally accomplished through automated, electronic means, it involves such practices as recording, authenticating, maintaining, and sharing accurate records of products. Outside-the-Box Benefits In addition to tightening up the supply chain and…
eBook: Of Microbrews and Medicines — Understanding Their Similarities and Differences in Bioprocessing Can Help Improve Yields and Quality While Reducing Cost
Meeting a biopharmaceutical scientist or engineer who proclaims a love for brewing is not surprising. Perhaps it’s because of the challenge of mixing raw ingredients together and waiting patiently for the final product, maybe it’s the hands-on nature of the equipment or the data analytics entertainment, or it just might be the simple joy of creating something. Whatever attracts a scientist or engineer to making medicines and/or craft brews, a surprising number of principles hold true for both bioprocesses despite…
eBook: Bioinks for Bioprinting — Three-Dimensional Printing in Research and Medicine
Three-dimensional (3D) printing is one method of digital biomanufacturing for both basic biological research and translational, clinical applications. The medical field has used it to create such constructions as 3D surgical models for preoperative planning, to assist surgeons in their procedure preparations, which improves postsurgical outcomes. Examples here include generation of cleft-palate models (1), orthopedic applications (2), and cardiovascular surgical planning (3). Other forms of 3D printing for biological applications — such as 3D bioprinting — go beyond such surgical…
eBook: Challenges Facing Biosimilar Entries into US Markets
Since the 2009 enactment of the Biologics Price Competition and Innovation Act (BPCIA) (1), the US Food and Drug Administration (FDA) has licensed six biosimilar products under PHS 351(k) and approved one product under FD&C 505 (b)(2). It also provided complete response letters (CRLs) to four biologics license application (BLA) filings (Table 1) (2). By comparison, the European Medicines Agency (EMA) has approved 31 biosimilar products (3) and refused or withdrawn about five. There is no doubt that US market…
Serialization: Background, Justification, Requirements, Timelines, and Readiness Across the Supply Chain
Drug manufacturers are facing unprecedented serialization challenges. Serialization requires weighty consideration and focused strategy for successful commercialization, even for those companies that have yet to bring a product to market. The World Health Organization estimates that 10% of medicines worldwide and up to 50% of drugs consumed in developing nations are counterfeit. In response to increasing drug integrity concerns, more than 40 countries have introduced laws mandating serialization and tracing of pharmaceutical products as they pass through the supply chain.…
Implementation of the BPOG Extractables Testing Protocols: Working with Multiple Single-Use Components
Single-use technologies offer significant advantages over traditional stainless-steel solutions for biopharmaceutical manufacturing. Reductions in setup times, cleaning and cleaning-validation costs, elimination of cross-contamination risks, and smaller footprints are just some of the benefits they provide. Although adoption of single-use systems (SUS) for commercial manufacturing is expanding, concerns persist that extractable and leachable (E&L) compounds from plastic SUS components potentially can leach into final drug products and compromise efficacy and safety. Those concerns are magnified amid the growing number of SUS…
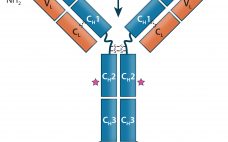
Therapeutic IgG-Like Bispecific Antibodies: Modular Versatility and Manufacturing Challenges, Part 1
Antibody-based immunotherapy has advanced significantly since 1986, when the US Food and Drug Administration (FDA) approved the first mouse monoclonal antibody (MAb) for clinical use: Orthoclone OKT-3 (muromonab-CD3). In the intervening years, researchers have applied the tools of genetic engineering to clone immunoglobulin G (IgG) genes into a number of expression vectors. In the 1990s, the bioprocess industry was able to produce fully human antibodies in cultured cells. As of June 2017, the FDA and the European Medicines Agency (EMA)…