Both small and large biopharmaceutical companies are increasingly pursuing the outsourcing of manufacturing and testing throughout the product lifecycle. The growing use of contract manufacturing organizations (CMOs) and contract testing organizations (CTOs) has led to increasing complexity within the biopharmaceutical industry as more third-party sites are leveraged to support global markets. To address those issues, a CASSS Chemistry, Manufacturing, and Controls (CMC) Strategy Forum was held in Washington, DC, 27–28 July 2014. The title was “Effective Management of Contract Organizations: Sponsors, Contract Organizations, Health Authorities and Patients — Keeping the Product Pipeline Moving, Compliant, and Available.” The CMC Strategy Forum is…
September 2015
From the Editor
We have had a busy summer, first adding to our annual Yearbook issue a supplement summarizing the well- attended presentations from our BioProcess Theaters at the Interphex and BIO events. And this month you are receiving our regular issue and annual guide to the upcoming BPI Conference and Exposition (25–29 October in Boston, MA) along with a sponsored supplement introducing cell-therapy initiatives at Pall Life Sciences and the third part of our special- report series featuring CMC Strategy Forum consensus…
September 2015 Spotlight
Sartorius Opens New Applications Center In June 2015, Sartorius opened an application center at its North American headquarters in Bohemia, NY. Among the guests were representatives from local government, customers and business partners, and industry media including BPI’s publisher, Brian Caine. The New York Sartorius Application Center provides customers with access to multiple single-use systems for upstream and downstream processing. Demonstration laboratories present equipment by application and to show how individual systems work together for complete process solutions. This 40,000-ft2…
Comprehensive Hands-On Training for Biopharmaceutical Manufacturing: BTEC’s Program to Deliver Training to FDA Investigators
Training and continuing education play a vital role in carrying out the US Food and Drug Administration’s mission to protect and promote the public health — not only for consumers, health professionals, and industry, but also for the agency’s own personnel. Since 2008, the Golden LEAF Biomanufacturing Training and Education Center (BTEC) at North Carolina State University has filled a niche in the agency’s internal training program and provided a series of courses to more than 100 FDA investigators. The…
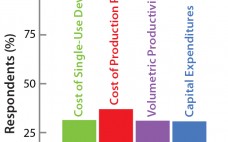
Bioreactor Design for Adherent Cell Culture: The Bolt-On Bioreactor Project, Part 4 — Process Economics
The Bolt-on Bioreactor (BoB) project is an independent initiative developing and commercializing a bioreactor for efficient, automated culture of adherent cells for biopharmaceutical applications (1). After conducting thorough research on available culture systems for adherent cells, the BoB team believes that a successful alternative to existing devices must solve four major challenges: volumetric productivity (2), process automation (3), containment and sterility (4), and process economics. This month concludes a four-part series addressing each of those challenges while describing design features…
Analysis and Characterization: The CMC Strategy Forum Series, Part 3 — Introduction
The CMC Strategy Forums provide a venue for biopharmaceutical product discussion. They focus on relevant chemistry, manufacturing, and controls (CMC) issues throughout the life cycle of a therapeutic and thereby foster collaborative technical and regulatory interaction. Forum chairs share information with regulatory agencies to help them merge good scientific and regulatory practices. Outcomes of the forum meetings are published in BioProcess International and on the CASSS website (www.casss.org). This process is meant to help ensure that biopharmaceutical products manufactured with…
Uniting Small-Molecule and Biologic Drug Perspectives: Analytical Characterization and Regulatory Considerations for Antibody–Drug Conjugates
Cosponsored by CASSS (an international separation science society) and the US Food and Drug Administration (FDA), the January 2010 CMC Strategy Forum explored antibody–drug conjugates (ADCs), which are monoclonal antibodies (MAbs) coupled to cytotoxic agents. The ADC platform of products is being used more and more for clinical evaluation in oncology. More than a dozen companies are developing several types, including products conjugated with calicheamicin, auristatins, and maytansinoids. Such products use the specificity of a MAb to deliver a cytotoxic…
Glycosylation of Therapeutic Proteins: Current Understanding of Structure–Function Relationships
A CMC Strategy Forum held in Washington, DC, on Sunday 28 January 2007 focused on two topics related to protein structure and function (1). First, analytical techniques used in the glycan analysis characterization included recent advances and correlations among the various tools. And second, current understanding of glycosylation’s functional relevance to therapeutic proteins was discussed in the context of its effects on biological activity, pharmacokinetics, and Fc effector functions (for monoclonal antibodies, MAbs). Progress has been made in the field…
Analysis and Structure Characterization of Monoclonal Antibodies
On 6 January 2003, 129 attendees participated in the second Well-Characterized Biotechnology Product (WCBP) Chemistry and Manufacturing Controls (CMC) strategy forum, titled “Analysis and Structure Characterization of Monoclonal Antibodies (MAbs),” held in San Francisco to discuss lot release and characterization test issues specific to MAbs (1). The objective of the meeting was twofold: to identify a “core” set of assays most useful for lot-release testing of MAbs and to define a mechanism for selecting appropriate potency tests. Two separate workshops…
Lot Release and Characterization Testing of Live-Virus–Based Vaccines and Gene Therapy Products
The January 2005 CMC Strategy Forum was devoted to a discussion of live virus vaccines and viral vectors used for gene therapy. The purpose of the meeting was to determine whether consensus positions could be reached among the delegates regarding lot release, stability, characterization, and comparability testing. Part 1 of this two-part report on that meeting describes factors influencing the choices of lot-release assays for vaccines and gene-therapy products (1). Part 2 presents potency testing, characterization, and comparability studies, including…