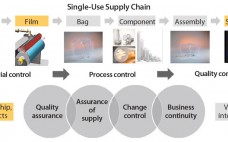
Growing adoption of single-use bags in commercial production of biopharmaceutical drugs raises new challenges for bag suppliers and drives the need for consistent product quality, improved assurance of supply, robust change management, and business continuity planning. In close collaboration with resin and film suppliers, polymer scientists and biologists at Sartorius Stedim Biotech have followed a stringent material science and quality by design (QbD) program to develop a completely new polyethylene film and to achieve consistent performance of new Flexsafe bags…
Manufacturing
Total Solutions Support the Growth of a Dynamic Industry: A Conversation with Reinhard Vogt and Stefan Schlack
While attending a conference at Sartorius Stedim Biotech in Göttingen, Germany, BPI publisher Brian Caine and editor in chief Anne Montgomery spoke with Reinhard Vogt (executive vice president of marketing sales and services, and member of the administrative board) and Stefan Schlack (senior vice president, marketing and product management). They discussed Sartorius’s forward-thinking business strategies, its position as a total solution provider, and how the company’s strategic goals mesh with its assessment of current industry directions. Single-Use As an Enabling…
Disposables for Biomanufacturing: A User’s Perspective
The supply scenario for many biopharmaceutical drugs such as monoclonal antibodies (MAbs) is changing. With the implementation of personalized medicine resulting in drugs for specific, high-responder subsets of patients, market volume per drug will decrease. In addition, increasing fermentation titers of up to 10 g/L for MAbs are leading to smaller fermentation volumes necessary to accommodate individual biopharmaceutical market demands. That results in approaches such as flexible production in campaigns or decentralization of manufacturing using similar facilities with low risk…
A Critical Mission: Clinical Trial Material Storage and Distribution
As if manufacturing of investigational medicinal products (IMPs) weren’t challenging enough already, the appropriate storage and distribution of sensitive biological products can be an adventurous journey itself — especially if not carefully planned and managed. No one solution fits all situations. Many things must be evaluated during planning stages. For example, what is more important: time to delivery or quality of transport and product integrity until drug can be administered to a patient? It is important to understand key storage…
BPI Theater at the 2014 BIO Convention
When we launched the BioProcess Theater series at the Biotechnology Industry Organization’s International Convention in 2007, we hoped that our special programming would fill a need within that event’s exhibit hall. We wanted to bring into the hall the type of technical presentations that are not generally part of the main event’s more executive-level, business-focused programming.It has therefore been especially gratifying to see our attendance growing every year — such that standing-room-only is becoming more the rule than the exception.…
Fed-Batch Cell Culture Process Development: Implementing a Novel Nutrient Additive for a Robust, High-Titer, Scalable Process
The fed-batch culture of Chinese hamster ovary (CHO) cells has become well established as the primary method of manufacturing therapeutic recombinant protein products for various disease indications. Fed-batch process-development approaches focus on supporting high–cell-density cultures that are crucial to achieving high product titers but lead to proportionately high nutritional demands. Exhaustion of key nutrients negatively affects cell growth and ability to produce recombinant proteins. To counter that problem, concentrated feeds are added to the culture. Such feeds tend to be…
UV-Vis Based Determination of Protein Concentration: Validating and Implementing Slope Measurements Using Variable Pathlength Technology
No longer are scientists bound to the time-consuming, error-prone use of dilution factors and fixed-pathlength measurements in determining the concentration of an analyte in solution. Using the slope spectroscopy technique, the Solo VPE system (from C Technologies) offers a new method of determining analyte concentration based on the Beer–Lambert law and slope derived from absorbance measurements made at multiple pathlengths (1). Mathematics: The Beer–Lambert law is expressed as A = αlc, where A is the measured absorbance, α is the…
Expansion of Human Mesenchymal Stem Cells: Using Microcarriers and Human Platelet Lysate
Cell therapy holds the promise of delivering the next generation of future medical breakthroughs. In this respect, multipotent progenitor cells such as human mesenchymal stem cells (hMSCs) have attracted high clinical interest because of their ability to differentiate into various cell types and their immunoregulatory properties. Furthermore, hMSCs express only low levels of class I major histocompatibility complex (MHC I) molecules on their surfaces and are therefore invisible to a host’s immune system. Finally, hMSCs can actively suppress the innate…
Biopharma Unhappy with Suppliers’ Progress on Single-Use Hookups
Single-use equipment vendors are failing to meet their biomanufacturing customers’ expectations in several key areas, according to BioPlan Associates’ 11th Annual Report and Survey of Biopharmaceutical Manufacturing Capacity and Production (1). The study — based on a global survey of 238 global biomanufacturers and contract manufacturing organizations, as well as 158 vendors and suppliers in 30 countries — identifies significant gaps between the perceived importance of key vendor attributes and companies’ satisfaction with their vendors regarding those attributes. One area…
Setting the Standard for Plasmid DNA Production
Boehringer Ingelheim stands out due to global contract manufacturing excellence in plasmid DNA production. It is one of the world’s leading companies for contract development and manufacturing of biopharmaceuticals