Once an engineering curiosity and smallscale laboratory technique, continuous bioprocessing has evolved in just a few short years to a topic of intense and increasing interest to most bioprocessors. Critics point to a steep learning/adoption curve, but that is nothing new in biomanufacturing.Andrew Zydney is a distinguished professor of chemical engineering at Pennsylvania State University. He has noted these challenges facing continuous processing: commercially unproven unit operations (especially downstream), a lack of equipment robustness, sterility concerns, and uncertain development timelines…
Manufacturing
Validation of Controlled Freezing and Thawing Rates: A 16-L–Bag Study
It is well understood that freeze– thaw processes affect the product quality of biopharmaceuticals (1–3). It has been reported that there is no consistent method of controlled freezing and thawing rates for biological formulations (4). Traditionally, ultralow temperature storage chambers that were not designed for freezing have been used to provide an energy state for the environment surrounding the product with very little excess capacity to change the state of the product. This study details a consistent method for controlled-rate…
Factories of the Future: Can Patient-Specific Cell Therapies Get There from Here?
In many ways, patient-specific cell therapies (PSCTs) are still the “new kid on the block” in medicine. Researchers, therapeutic developers, manufacturers, regulators, and payers are still exploring and developing an understanding of the powerful benefits and unique challenges associated with this growing industry. As we all become more familiar with PSCTs, an evolution will need to occur — as it has for automobiles, computers, and every technological advance in human history — for these therapies to become widely adopted, cost-efficient,…
Emerging Platform Bioprocesses for Viral Vectors and Gene Therapies
Recent advances in molecular biology are expediting genomic sequencing to underpin precision medicine. Such progress is positioning gene and gene-modified cell therapy on the cusp of an extraordinary revolution in patient care for presently unmet medical needs — and a new therapeutic class that could rival monoclonal antibodies (MAbs) in importance. However, despite substantial strides made in clinical trials, the bioprocessing community is struggling to fulfill growing demands for biomanufacturing capacity to make gene and gene-modified cell therapies — including…
Cancer Immunotherapies: Fulfilling the Promise of Protein and Cell Therapies
With few exceptions, both small-molecule and biological cancer treatments have contributed only incrementally towards achieving long-term responses or outright cures. In this regard, emerging cell- and protein-based cancer immunotherapies represent game-changing strategies for treating even refractory cancer. With long-term responses now possible, medical science may be on the verge of delivering on the long-unfulfilled promise of making cancer a manageable disease. But impediments to commercializing cancer immunotherapies are substantial. Producing cell-based treatments entails substantial hands-on manipulation and perfecting the logistics…
Automation of CAR-T Cell Adoptive Immunotherapy Bioprocessing: Technology Opportunities to Debottleneck Manufacturing
Continued clinical efficacy demonstrations of cell-based immunotherapies (iTx) such as chimeric antigen receptor T cell (CAR-T) therapies has made the prospect increasingly likely of an immunotherapy product achieving conditional market authorization in the short term. For example, Novartis and the University of Pennsylvania’s lead candidate (CTL019) for treating a range of hematological malignancies received breakthrough status from the US Food and Drug Administration (FDA) in 2014, permitting access to an expedited drug development pathway for high unmet medical needs (1).…
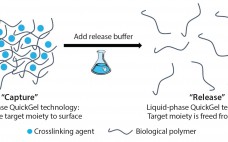
Development of a Novel Cell-Separation Platform: Discussion with Quad Technologies CEO Sean Kevlahan
Releasing and separating cells from surfaces and capture molecules are critical steps in cell therapy development. Research into such therapies as chimeric antigen receptor T cells (CAR-T) cancer therapies and stem-cell regenerative medicines demand the isolation and purification of viable and functional target cells. A number of cell-separation strategies can be used to produce such cells, but they are not able to deliver the required efficiency or scalability and can also cause damage to cells or affect their phenotype. Since…
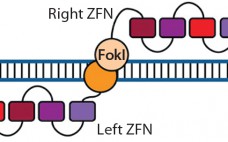
Therapeutic Gene Editing: Tools to Facilitate Basic Science or Stimuli for a Paradigm Shift in Biomanufacturing?
Historically, fundamental science and process engineering were separated by distinct vernaculars and a decade or more in the translation pathway of candidate therapeutics from laboratory to bedside (1). This crude metric holds true for the origins of the modern pharmaceutical industry, namely fine chemicals that supported the high-margin small molecules that constitute the majority of the pharmacopoeia even today. But as illustrated by deeply interwoven careers, companies, and technologies — including those related to monoclonal antibodies (MAbs) — that classic…
Bioprocessing Standards for Single-Use Components Are Moving Forward
With the escalating use of single-use technology in bioprocessing, suppliers have had to rapidly develop disposable components such as fittings, tubing, pumps, sensors, and flexible containers for bioprocessing. Single-use technology is growing so fast that the organizations tasked with guiding its growth are having difficulty keeping up. Contributing to this problem are factors such as company needs, regulatory requirements, market pressures, and costs. That growth has posed considerable concerns for the bioprocessing industry about the presence of organic and inorganic…
Rapid Formulation Development for Monoclonal Antibodies
Monoclonal antibodies (MAbs) are at the focal point of biologics development. Many of the best-selling drugs are therapeutic MAbs or related proteins (1–2). The combined world-wide sales from MAbs will be nearly US$125 billion by 2020 (3). About 50 MAb products treating a range of diseases have been approved in the United States or Europe. With the large number of MAbs progressing through discovery, biomanufacturers need to accelerate process development and move projects rapidly into clinical manufacturing (4–5). Formulation development,…