The implementation, maturation, and benefits of single-use technologies in biopharmaceutical development and manufacturing are well documented and understood. As analytical methods and testing services also rapidly improve, it is clear that management of risk associated with extractables and leachables also must evolve. Standardization is universally accepted as a goal; how to define, implement, and educate the industry is where debate resides. The container–closure segment has had more experience dealing with leachables and extractables than those implementing single-use process components do…
Manufacturing
Drivers, Opportunities, and Limits of Continuous Processing
Biologics represent a significant group of approved drugs, and they are expected to continue their exceptional growth in the foreseeable future. At first glance, innovative monoclonal antibodies (MAbs) and their derivatives seem to dominate the field; however, biobetters, biosimilars, and other therapeutic proteins have gained in relevance lately. This diverse portfolio also is reflected in the range of different biomanufacturing processes used. Most blockbuster antibodies are produced by mammalian cells cultured in large bioreactors (10–20 m³) in fed-batch mode. But…
Accelerated Development Through Strategic Analytical Partnerships
The analytical field for biologics has evolved greatly over the past 30 years, and the underlying growth has shifted from biopharmaceutical companies to contract research organizations (CROs). The global biopharmaceutical market is growing annually at >15%, making it the largest and consistently fastest growing segment of the healthcare industry with annual sales in excess of US$200 billion. Contract manufacturing organizations (CMOs) are expanding capacity by building new cost-efficient facilities, reflecting market demand. Many product sponsors are outsourcing, some even increasing…
Outsourcing Biosimilar Development
As the debate continues over the high cost of pharmaceutical treatment options, the development of biosimilars continues to play a dominant role in that discussion and will be an important part of the solution. Biosimilar companies are working at a feverish pace to develop the next generation of follow-on products. Outsourcing to a growing group of contract development and manufacturing organizations (CDMOs) is a key strategy for savvy developers to accelerate their products’ launch. Finding the right CDMO isn’t an…
Outsourcing Manufacturing and Analytical Needs in the Biopharmaceutical Industry
Expanding biopharmaceutical pipelines and therapeutic modalities are feeding a boom in outsourcing key elements of development and manufacturing projects. The rapid growth of the industry as well as of emerging therapeutic areas (e.g., antibody–drug conjugates, biosimilars) challenge sponsors and contractors alike to meet industry needs. The authors in this featured report explore increasing complexities of devising long-term business strategies, the importance of choosing between long-term partnerships or one-time/transaction-based projects; the assurance that adequate business support and technical capabilities exist on…
Critical Aspects of Technology Transfer to CMOs
Technology transfer in pharmaceutical manufacturing most often is mentioned as occurring between two organizations and involving discrete product lifecycle stages: e.g., a sponsor and a contract manufacturer and from development to manufacturing. According to the International Council on Harmonization of Technical Requirements for Pharmaceuticals for Human Use: “The goal of technology transfer activities is to transfer product and process knowledge between development and manufacturing, and within or between manufacturing sites to achieve product realization. This knowledge forms the basis for…
Specialized Outsourcing Services: Antibody–Drug Conjugates
Managing just the conjugation part of an antibody–drug conjugate (ADC) project can be a challenge. So when you consider all the component parts of an ADC, it is understandable why so many related activities are outsourced. At Piramal Grangemouth, we provide conjugation development and manufacturing services. Together with our sister sites in the United States and United Kingdom, we offer payload manufacture and fill–finish capability. The Grangemouth (UK) site has been involved in ADC conjugation services now for over 13…
Introduction: Process Issues in Cell, Gene, and Tissue Therapies
It’s hard to believe that just six years ago, BioProcess International published its first cell therapy supplement, which included just one article on “cell therapy bioprocessing” (1). At the time, most such processing was conducted in special clinical laboratories and academic institutions. As BPI continued to cover this relatively new segment of the biopharmaceutical industry, we heard more about “the product is the process” and “scale out instead of scaling up.” After many trials, errors, and milestones, regenerative medicine has…
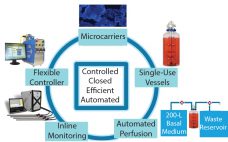
Expansion Platform Components
I first met Chris Mach at the Biotech Week Boston conference in October. We discussed the challenges that biomanufacturers are facing in cell expansion, especially in three specific areas in scale-up systems. View the full article below – Login Required
Platform Solutions for Cell Therapy Manufacturing
Advances in cell therapy have resulted in significant progress toward treating some widespread and difficult diseases, many of which represent unmet medical needs. For example, phase 3 clinical trials are already under way for therapies based on mesenchymal stem cells (MSCs), including therapies for graft-versus-host disease, acute myocardial ischemia, and chronic obstructive pulmonary disease (COPD) (1–3). Successful cell therapy treatments for such afflictions will be not only significant medical breakthroughs, but also in very high demand. However, their commercialization is…