In late October 2018, KNect365 brought together more than 250 analytical specialists to discuss characterization of well-characterized biologics in Rockville, MD. Speakers from the US Food and Drug Administration joined experts from leading biopharmaceutical companies, service providers, and consultancies, including BPI editorial advisor Nadine Ritter (president and analytical advisor of Global Biotech Experts). She began the final day moderating a special town-hall session where audience members could pose their regulatory questions to a panel of FDA reviewers, and she ended…
Analytical
Biopharmaceutical Characterization,
Speeding Characterization of Biologics: Replace Traditional Assay Technologies with Label-Free Quantification and Kinetics
FortéBio’s Octet instruments are an ideal replacement for ELISA, HPLC, and SPR techniques in quantification of antibodies and recombinant proteins and in testing product potency for lot release. Bio-Layer Interferometry (BLI) technology monitors biomolecular interactions in real time to determine affinity, kinetics, and concentration. The plate-based, microfluidics-free format offers users several distinct advantages over other technologies. BLI-based systems can achieve higher throughput, with the flexibility to measure two to 96 samples simultaneously. Lower maintenance requirements and increased ease-of-use further shorten…
Multitiered Automation for Improved Efficiency of Bioprocess Analytics
The first biopharmaceutical, human insulin, was approved for use in 1982 (1). The biopharmaceutical market continues to exhibit healthy growth now, with the number of yearly patent applications increasing by 25% annually since 1995 (2). The total pharmaceutical R&D pipeline has more than doubled since the beginning of the century (Figure 1), much of that attributable to the biologics industry segment. As this industry has matured, new platform methods have emerged, and competition has increased. Consequently, the pressures of speed,…
eBook: Using Modern In Situ Analytics and PAT for Automated Feedback Control of Critical Process Parameters
Simply put, the best way to control a critical process parameter (CPP) is to measure that specific parameter, integrate the live signal into your control system, and apply a smart feedback algorithm for an automated control loop. The challenge in doing this for bioprocesses has been due, in part, to the complex, highly dynamic, and variable nature of the process along with the lack of robust, scalable, and multiformat (single-use or multiuse) technologies that can monitor in real time such…
Viral Risk Mitigation: A Global Regulatory Perspective
The production of biologics will always have the risk of viral contamination. Manufacturers have developed a multitiered approach — tailored to individual processes — to prevent adventitious viruses from entering production processes, detect contamination in raw materials and process intermediates, and remove viruses in downstream purification. This article provides an overview of the global regulatory framework to ensure the viral safety of biologics. Past Contamination Events Past contamination events have resulted in corrective and preventative actions to reduce the risk…
A Novel 3D Culture System for High-Throughput Hepatoxicity Screening
Cells grown as three-dimensional (3D) spheroids are thought to more closely mimic in vivo physiology in terms of morphology, structural complexity, and phenotype. Being more physiologically relevant, 3D cultures can be highly predictive for compound profiling and evaluating cytotoxicity, a critical step in evaluating chemotherapeutic drug candidates. Unfortunately, evaluation of drug cytotoxicity traditionally has relied on the use of two-dimensional (2D) cell culture monolayers. When grown in monolayers, cells are not exposed to soluble gradients, are forced into an apical-basal…
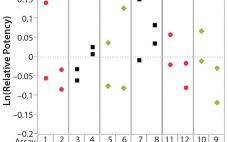
Certain Approaches to Understanding Sources of Bioassay Variability
During lifecycle development of a biological assay (bioassay), identifying and reducing sources of variability might be required to improve method performance. Here I recommend some statistical and graphical approaches (consistent with USP <1033>) for practitioners to identify variation from experimental results (1). Sources of Variation in a Bioassay To correctly identify sources of variation in a bioassay, analysts must consider how that bioassay is to be executed. In particular, the experience and technical expertise of each analyst expected to execute…
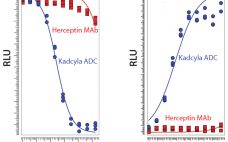
Biological Stealth Bombers: Potency, Regulatory, and Bioprocessing Concerns of Antibody–Drug Conjugates
Seven years ago, the US Food and Drug Administration (FDA) approved the first product in a new class of biologics: antibody–drug conjugates (ADCs). The idea for these products already had been hatched a decade earlier when the promising field of antibody research — touting such molecules as “magic bullets” — had faltered, specifically against oncology-related indications. The early crop of anticancer monoclonal antibodies (MAbs) proved to have only limited efficacy, and interest in developing antibodies as therapeutic agents against cancer…
Single-Use Sensors and Control and Data Acquisition Tools to Streamline Bioprocess Development
Process development and biomanufacturing in the biopharmaceutical industry have evolved extensively over the past 10 years. More tools are available to study process variables to enable more efficient and productive processes, speed development, and reduce costs. High powered microcontrollers are embedded in laboratory devices to carry out complex tasks. Recently, users have started working with microcontrollers such as Raspberry Pi for personal projects. As personal computer power has accelerated multiplefold,leading to high processing power and compact, high-capacity memory readily available…
Trends in Chemistry, Manufacturing, and Controls: Next-Generation Technologies and Product Modalities
New technologies bring new regulatory challenges. The biopharmaceutical industry must be cautious in its implementation of new scientific ideas and technology platforms — no matter how promising those might be. Regulators will look skeptically on any claim that isn’t backed up by good data, and with no solid history of successful use to build on, a company must have all the answers itself. How do compliance professionals anticipate what kinds of questions reviewers will ask when the time comes —…