This webcast features: Joe Ferraiolo, Associate Director, Bioanalytics Applications, C Technologies, a Repligen Company Analytical technologies play a crucial role in accelerating gene therapy speed to market. Dilution steps during protein and DNA quantitation measurements represent the single highest source of accuracy and precision errors. The SoloVPE® device provides a dilution-free workflow that saves time, reduces deviations, and facilitates multisite alignment. Multiple case studies from major biopharmaceutical companies describe execution in process development, analytical labs, and directly on good manufacturing…
Manufacturing
Figure Out Any Particle with Automated Counting, Sizing, Morphology, and Identification Using the Hound
This webcast features: Lisa Krapf, Field Automation Scientist, Unchained Labs Thorough particle sizing and analysis are essential steps in formulation development and the monitoring of manufacturing process controls. Traditional particle analysis techniques such as light obscuration do not provide morphological information that can be useful for classification. Transfer to manufacturing and scaling to production will necessitate different equipment and the potential introduction of extrinsic particles that may not have been observed previously. The combination of particle analysis with flexible options…
Protein or Not? Advanced High-Throughput Aggregate Analysis with the Aura™
This webcast features: Bernardo Cordovez, Chief Science Officer and Founder, Halo Labs In protein-based formulations, distinguishing aggregated active pharmaceutical ingredient (API) from other particle types is important for understanding the root cause of instability. Until now, existing methods have been either unreliable or too cumbersome to use in many workflows. Here we introduce the Aura™ 96-well low-volume aggregate and particle imaging system, which can rapidly size, count, and characterize particles and identify them as proteins, non-proteins, hydrophobic, or other types…
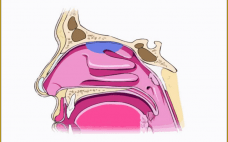
Translating Inhaled and Nasal Technologies for the Delivery of Biologics
This webcast features: Mark Parry, Technical Director, Intertek Inhaled and nasal delivery platforms have specific applications outside of their traditional uses for asthma/chronic obstructive pulmonary disease (COPD) and seasonal rhinitis/sinusitis: They can offer real advantages for the delivery of therapeutic biologics. During this short presentation, Intertek’s Technical Director, Mark Parry, will provide an overview of currently available technologies and successfully marketed products, with a look at the development challenges that might be encountered — and the solutions that are available…
Relative and Absolute Quantitation of Impurities and HCPs Using Mass Spectrometry
This webcast features: Steven Broome, Senior Mass Spectrometrist, BioPharmaSpec The processes involved in manufacturing a biopharmaceutical use biological and chemical systems to produce and purify the drug product. Therefore, the final active pharmaceutical ingredient (API) will often contain impurities related to these processes. It is a regulatory requirement to identify and monitor process-related impurities, and a qualitative and quantitative assessment of the components in the final drug product must be performed. Knowledge of the product-specific impurities, such as host-cell proteins…
Optimizing Your Excipient Screening for Vaccine Formulation with an Ultra-Pure Pharmaceutical Gelatin
This webcast features: Jeroen Geeraerts, Business Development Manager, Biomedical, Rousselot The world is working at an unprecedented pace to develop a safe and effective vaccine to combat the COVID-19 pandemic. Currently, five candidate vaccines are in clinical evaluation, and many more are in preclinical testing. Different types of vaccines are being developed using multiple strategies and platforms. Among them are several inactivated-virus and live-attenuated–virus candidate vaccines. As an excipient, gelatin is a key component in many vaccine formulations. Well-known examples…
Technology-Ready Processes for Gene Therapy Manufacturing 2.0
This webcast features: René Gantier, PhD, Director of Technology, Gene Therapy, Repligen The current manufacturing processes for viral vectors for gene therapy, which we can define as Gene Therapy Manufacturing 1.0 (e.g., adherent cell culture and transient expression from plasmid transfection), are not productive enough to meet the future demand considering the quickly increasing number of approved gene therapies and clinical trials. A transition is therefore ongoing to implement more productive and scalable processes, leading to Gene Therapy Manufacturing 2.0 using…
Innovative Closed Process CAR-T Cell Therapy Platform to Streamline Approach for Manufacturing with Great Predictability
This webcast features: Tatiana Golovina, Senior Director, Cell Therapy Process Development, WuXi Advanced Technologies For many years, the primary forms of cancer treatment have been chemotherapy, radiation, and surgery. An amazing breakthrough known as chimeric antigen receptor (CAR) T-cell therapy is being studied in the treatment of various types of cancer, including acute and chronic lymphoblastic leukemia, non-Hodgkin lymphoma, myeloma, and solid tumors. Developing innovative advanced therapies is one of our greatest opportunities to dramatically improve patients’ lives. WuXi Advanced…
World-Class AAV Vector Suspension Platform to Accelerate Commercialization to Market with Greater Predictability
This webcast features: Juan Lagos and Steve Tottey, Associate Directors of Upstream and Downstream Process Development, WuXi Advanced Therapies Developing innovative advanced therapies is one of our greatest opportunities to dramatically improve patients’ lives. WuXi Advanced Therapies launched a new world-class adenoassociated virus (AAV) vector suspension platform that complements integrated capabilities enabling cell and gene therapies to be developed, manufactured, and released faster and with greater predictability globally. In this webinar, we will discuss our recent technical advancement on efficient…
Assembly, Labeling, and Packaging Trends: Next Decade’s Sterile Finished Goods Solutions
This webcast features: Thomas Gabriel, Director, Strategy and Business Development, FUJIFILM Diosynth Biotechnologies Learn new trends in packaging, labeling, and distribution of finished goods and supporting technology platforms. At this event, there will be a presentation and dialogue regarding: patient convenience: trends, approaches and demands for future treatments medical device delivery systems of the next decade digitalization’s improvements to patient quality of life and medicinal ease of use manufacturing and supply chain networks of the future. Watch the recorded webcast…