Sean Liour (vice president for project management at GenScript ProBio) delivered an Ask the Expert presentation on 18 March 2020 to explore the advantages of his company’s platform for cell-line development. Liour explained that successful biologic development hinges on robust host cell lines, capable expression vectors, and discriminating clone-screening systems. Overviewing relevant technologies and capabilities, Liour illustrated how his company’s ProCLD platform helps sponsors navigate cell-line development. Liour’s Presentation Researchers must weigh their options carefully when selecting a host cell…
Upstream Processing
Navigating Technology Transfer
Technology transfer is a key milestone in the journey from discovery to full-scale good manufacturing practice (GMP)-compliant manufacturing. Navigating this step while preventing unforeseen issues that can create costly delays is supported best by combining knowledge of a given process with understanding of the technological capabilities. Different applications have different needs. Some challenges and goals are common to bioreactor processes for suspension and adherent cell culture for production of viral vectors, monoclonal antibodies (MAbs), other recombinant proteins, and vaccines. All…
Industrialize Your Viral Vector Production in Adherent and Suspension Cell Cultures: Know the Pros and Cons
This educational podcast, “The Evolution of Culture Systems for Viral Vector Production: Advantages, Challenges and Cost Considerations,” recently published by Cell and Gene Therapy Insights, discusses in detail the pros and cons of viral vector production in adherent and suspension cell culture. This special report illustrates how Pall Biotech’s iCELLis 500+ bioreactors and Allegro STR bioreactors can bolster adherent and suspension culture, respectively, for viral vector production. Fill out the form below to read the complete report and learn more now.…
BVDV Risk Mitigation: Dealing with Bovine Viral Diarrhea Virus in Serum
Bovine serum products such as fetal bovine serum (FBS) are critical, nutrient-rich supplements frequently used in cell culture systems for a number of applications, including biotechnology, animal and human pharmaceutical and diagnostic manufacturing, and life-science research. Serum can be contaminated with adventitious agents that could increase its risk for use in cell culture systems. Bovine viral diarrhea virus (BVDV) is one of the most significant infectious diseases in the livestock industry worldwide because of its high prevalence, strong persistence, and…
Brazilian JBS Federally Inspected Fetal Bovine Serum
Serum is the most commonly used supplement in cell culture. Fetal bovine serum (FBS) is the common choice because it contains high concentrations of growth factors and other important signaling molecules (e.g., adhesion proteins, nutrients, carrier proteins, cytokines, and hormones) required for cell survival and differentiation together with its buffering capabilities. FBS production begins with the collection of whole blood from bovine fetuses under aseptic conditions. Once collected, the blood is allowed to clot, and the serum is mechanically separated.…
eBook: Microbial Expression — The Right Choice for Large Peptides and Small Proteins
Although animal cell culture has dominated the biopharmaceutical industry for some years now, microbial expression remains important for producing proteins that don’t require posttranslational modifications — or only those that prokaryotic microbes can perform. It also offers an affordable option for antibody fragments and gene therapies. Microbes may be less fragile than animal cells, and they do require simpler media, but they present other challenges related to temperature management and oxygen transfer in culture. Wherever practical, bacterial expression is preferred…
Implementation of Single-Use Miniature Bioreactors to Support Intensified Cell Culture: Using Functional Performance Indicators to Assess a Small‑Scale Model
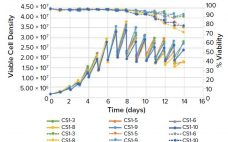
Changes to bioprocessing in the biopharmaceutical industry are driven by the need for increased speed, lower cost of goods (CoG), and greater flexibility (1). To meet these challenges, the industry is adopting strategies that include intensified processing. During the initial stages of intensified processing, it is essential to identify the most productive and/or stable clones for use before starting pilot-scale studies. That requires screening large numbers of clones and then further testing the most promising ones in benchtop bioreactors. The…
Improving Bioprocess Expression Systems: A Clean Alternative to CRISPR/Cas9
Chinese hamster ovary (CHO) cells have emerged as a robust platform for bioprocessing serving both early and late-stage biotherapeutic drug supply. However, these cells and other hosts (e.g., HEK293), can be optimized for even greater potential through advanced gene editing. For example, when the endogenous glutamine synthetase (GS) gene is knocked out in CHO cells, a sixfold increase in high-producing cell lines is achieved (1). In another study, CHO with annexin A2 (ANXA2) and cathepsin gene (CTSD) knockouts were introduced…
Ask the Expert: Accelerating Timelines By Integrating Cell-Line Development and Manufacturing
In a 31 October 2019 “Ask the Expert” presentation, Nicole Wakes (group leader of Abzena’s cell-line development team) observed that drug sponsors often outsource their early upstream activities to a few different contract research organizations (CROs). But that strategy can thwart short timelines and introduce regulatory and financial risks. Wakes described Abzena’s upstream approach, illustrating how partnering with a single, multicompetent CRO from cell line construction through manufacture can streamline workflows. Integrating cell line development and manufacturing in this way…
Ask the Expert: Cell Culture Media Analysis Using Handheld Raman Analyzers
In biopharmaceutical manufacturing, cell culture media supply critical nutrients and maintain pH and osmolality to optimize protein product yield. Because media composition and condition have a strong effect on final biologic product quality and production, biopharmaceutical companies monitor media for lot-to-lot variability. Stability testing for degradation due to light exposure, temperature changes, or shelf-life/time is possible with rapid spectroscopic methods. In an 8 October 2019 “Ask the Expert” webinar, O. Dean Stuart (product manager at Thermo Fisher Scientific) explained how…