This webcast features: Ashleigh Wake, Business Development Director, Intertek Pharmaceutical Services Inhaled and nasal biologics continue to grow in importance as advantages presented both by targeted delivery to the lung and systemic delivery for other diseases or treatment pathways are driving this increased attention. In this webinar, Ashleigh Wake describes the top development considerations for inhaled or intranasal biologic analytical development. These include the need to meet regulatory requirements of ICH Q6B for a well characterized biologic as well as…
Sponsored Content
Standardized AAV manufacturing: Why are AAV reference materials needed?
The field of gene therapy is in great need for well characterized AAV reference materials, according to viral vector delivery services firm Vigene Biosciences. Recombinant adeno-associated virus (rAAV) is a widely used gene delivery tool for research and clinical applications in the gene therapy field. To compare the pharmacokinetics and efficacy of rAAV, well characterized AAV reference materials are needed for assays and in-house reference materials calibration. AAV reference materials are also essential for AAV in process development and AAV…
Simplifying the Bioprocessing 4.0 Journey
Bioprocessing 4.0, the biopharmaceutical version of Industry 4.0, is on course to become a reality in the next decade (1). This is because its “cyber-physical systems” that comprise cloud computing, connected systems, and digital process control offer many benefits. They include better process monitoring and management of a biologic’s critical quality attributes (CQAs) and the chance to control intensified processes for faster, less-expensive production of protein-based biologics and vaccines. Automation also can reduce the number of skilled operators needed while…
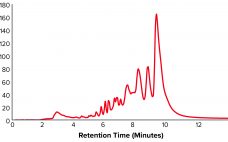
Development of a Universal Preparative Anion-Exchange Method to Purify Oligonucleotides
Oligonucleotide-based therapeutics have been studied over recent decades, and their promise as a new drug modality is now being realized. The growing interest in oligonucleotides is driven by their high potential for treating different medical conditions, the growing number of oligonucleotide drugs approved by the US Food and Drug Administration (FDA), an increased focus on personalized medicines, the development of therapies for rare diseases, and the wide adoption of nucleotide-based COVID-19 vaccines. Oligonucleotides are short, linear sequences of DNA or…
Strategizing Scale-Up and Scale-Out for Cell Therapy Production
When considering strategies for expanding the number of cells being grown to support cell therapy development, companies often focus on decisions regarding scale-up and scale-out: increasing capacity either by using larger vessels to increase production volume or by implementing more units of the same vessel, respectively. Complete workflows often involve both. Figure 1 shows an example of scaling out from one to multiple cell culture flasks of the same dimension before transitioning to a larger format. Scale-out can be straightforward…
The Effect of Benzonase Endonuclease Addition to Purification of Sabin Poliovirus Type 3
During production of vaccines and viral vectors, the size and quantity of extracellular nucleic acids must be reduced using endonuclease enzymes. Merck/MilliporeSigma’s proprietary Benzonase endonuclease is a genetically engineered nuclease derived from the Gram-negative bacteria Serratia marcescens. It attacks and degrades all forms of DNA and RNA. It is manufactured under good manufacturing practice (GMP) conditions and has a drug master file (MDF) in place with the US Food and Drug Administration (FDA), which can be cited in regulatory filings.…
Single-Use Technology for Formulation and Filling: A Case Study from Swissfillon AG and Pall Corporation
Swissfillon AG is a contract manufacturing organization (CMO) based in Switzerland. Fully compliant with current good manufacturing practice (CGMP) regulations, it provides state-of-the-art aseptic filling for pharmaceutical and biotechnology companies, from clinical-phase materials to commercial quantities. This CMO specializes in high-value, difficult-to-fill products. Swissfillon recognized that adoption of single-use systems (SUS) on a commercial scale required major improvements in consistency and reliability compared to manual operations at pilot and clinical-trial scale. The single-use formulation and filling process, which includes an…
GatorTM AAVX Probes for Rapid and Label-free Quantitation of AAV Serotypes
Adeno associated virus (AAV) capsids are a leading modality for in vivo gene delivery. Complete and precise characterization of capsid particles, including capsid and vector genome concentration is necessary to safely and efficaciously dose patients. In virus development and production, it is important to determine the virus concentration at different stages of the process, optimize the clone used and obtain best production yields. Easy, rapid and cost effective quantitation methods are critical to achieve above goals. The Gator AAVX probes…
Ask the Expert: Facilitating Process Development Using a Microfluidic Perfusion Bioreactor
Suitable scale-down perfusion systems generally have been unavailable for process development (PD) activities. Some commercially available systems require daily media exchanges. No such system performs in a way that accurately represents large-scale perfusion, and none maintains sufficiently high cell densities. Kevin Lee (cofounder of Erbi Biosystems) joined BPI on 4 May 2021 to explain how his company’s Breez bioreactor system integrates all the functions of a stirred-tank reactor (STR) into a compact format that can facilitate PD, enabling one person…
Ask the Expert: Streamlining Mesenchymal Stem Cell Production — From Expansion Through Removal of Cryoprotectant
The number of cell therapy product candidates based on mesenchymal stem cells (MSCs) has grown steadily since their clinical debut in 1995. As of June 2020, clinical investigators were evaluating more than 1,100 such therapies. Scaling up MSC production remains challenging, however. On 31 May 2021, Hilary Sherman (senior scientist at Corning Life Sciences) presented an “Ask the Expert” webinar describing her company’s efforts to facilitate MSC workflows. Sherman’s Presentation Easing Expansion: MSCs have strong differentiation capability and can be…