Making use of chromatography resins with better selectivity, resolution and capacity is one approach to solving the problem not only of aggregate removal, but also that of MAb fragment and other digest products, in a column chromatography purification step. The data presented here demonstrate the capabilities of CaPure-HA to separate Fab and Fc fragments from undigested MAb in a papain digest of an IgG1 monoclonal antibody.
Sponsored Content
Cadence™ Single-Pass TFF Coupled with Chromatography Steps Enables Continuous Bioprocessing While Reducing Processing Times and Volumes
Continuous bioprocessing initiatives have accelerated in recent years. The transition from batch to continuous bioprocessing offers numerous advantages, including process time savings and lower capital costs due to smaller equipment, tanks, and tubing sizes. Additionally, hold tanks may no longer be needed between unit operations, which can be especially valuable for facilities with limited manufacturing floor space. Furthermore, reduced system hold-up volumes improve product recoveries and also contribute to the smaller system footprint of continuous bioprocessing systems. One unit of…
Volume Reduction and Process Optimization with Cadence™ Inline Concentrator
Pall’s Cadence™ single-pass tangential flow filtration (SPTFF) technology provides a simple solution for continuous concentration and optimization of Downstream Processing (DSP) steps. It eliminates the conventional TFF recirculation loop and allows product to be concentrated in a single pump pass. The single-pass operation eliminates any mixing or foaming issues and exposes the product to low shear; thus, it is optimal for the processing of fragile and shear-sensitive molecules as well. The single-pass feed flow rates are lower compared to conventional…
Design of a New Range of High Capacity and High Flow Agarose Resins
With the continuing development of high titer cell lines, resins with very high capacity at relevant residence times are becoming increasingly important. This paper will present application and characterization data from a set of novel, agarose-based resins, with ion exchange functionality as well as with immobilized alkaline stable Protein A. The cation exchanger has a dynamic binding capacity of more than 120 mg monoclonal antibody/mL resin without the introduction of surface extenders. Performance data from an alkaline stable Protein A…
Production Scale Prepacked Chromatography for Use with 1000 L and 2000 L Bioreactors
This webcast features: Fletcher Malcom, Associate Director of Product Management at Repligen. The technical and economic benefits of ready-to-use, pre packed chromatography columns at bench- and small-scale have been proven and documented. The question remains: how do we scale up to production-scale? Now, for the first time, the new OPUS® 45 and 60 cm columns can purify a feed stream from a 1000L – 2000L bioreactor. This Ask the Expert webcast will show how the unique design of these GMP-scale…
Exploring Protein Stability by nanoDSF
nanoDSF determines thermal and chemical protein stability with ultra-high resolution and with unmatched reproducibility. The detection of the intrinsic tryptophan fluorescence enables measurements at close-to-native conditions in any buffer, even in the presence of detergents. nanoDSF is applied in antibody engineering, membrane protein research, formulation and quality control.
Single-Use-Bioprocesses – Hype or Future Technology?
Single-use technology have become indispensable in biopharmaceutical manufacturing. Rentschler is one of the first contract manufacturers worldwide to establish a complete single-use facility for the upstream and downstream processes.
A Xeno-Free Culture System for hMSC from Various Sources Suitable for Initial Isolation and Expansion Toward Clinical Applications
Human mesenchymal stem cells (hMSC) are multipotent adult stem cells present in a variety of tissue niches in the human body. hMSC have advantages over other stem cell types due to the broad variety of their tissue sources, since they are immuno-privileged, and for their ability to specifically migrate to tumors and wounds in vivo. Due to these traits hMSC have become desirable tools in tissue engineering and cell therapy. In most clinical applications hMSC are expanded in vitro before…
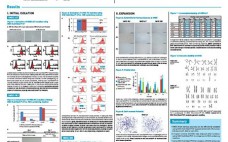
CaPure-HA™: DoE Optimization of Elution Conditions
Design of Experiments (DoE), also called experimental design, is a statistical approach to process development that has gained wide acceptance in the biopharmaceutical industry. DoE is used to reduce development costs by speeding up the design process and to optimize the parameters of a particular step in the manufacturing process. The data presented here demonstrate the optimization of the CaPure-HA elution using a DoE approach.
Pressure Interruptions (Stop/Start) During Virus Filtration: Assuring Safety Using Robust Process Technology and An Appropriate Risk Mitigation Strategy
Following publications discussing the impact of pressure interruptions on the retention levels of virus filters, an increased focus is being placed on filter users’ own risk mitigation strategies. Users are increasingly requesting technical guidance and support with viral validation from filtration experts, as part of their overall product support package. This guidance document provides a high level of assurance to filter users by demonstrating how combining robust virus clearance technology with the use of standard operating procedures can assure a…