Based on the many forms that modern vaccines can take, their manufacturing is complicated. Unlike monoclonal antibodies (MAbs), vaccine manufacturers have no “template” platform to follow. Most vaccine producers develop their manufacturing processes from scratch, a prospect that can be challenging for small to mid-sized companies. Bioprocessing is the key challenge in vaccine manufacturing. Without a well-developed and understood process, a manufacturer will face serious challenges in commercial production: e.g., low yields, high costs, and difficulties in meeting quality standards.…
Vaccines
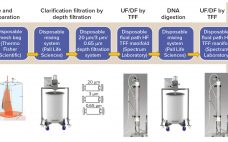
Adenovirus Downstream Process Intensification: Implementation of a Membrane Adsorber
Historically, companies developing vaccines have used attenuated pathogens, inactivated infectious agents, or antigenic constituents purified from pathogenic sources. In the past 20 years, technological advances such as recombination and viral vectors, have enabled development of vaccines against diseases with previously no available treatments (1). Viral vectors have become one of the most rapidly evolving and promising fields in vaccinology and regenerative medicine. In addition to preventing infectious disease, they have a broad range of potential applications, including treatment of hereditary…
Advances and Challenges in Vaccine Development and Manufacture
Scientists have made significant breakthroughs in bioprocess and analytical technologies for supporting vaccine development. Such technologies have helped vaccine manufacturers achieve consistent product purity and quality rapidly and cost effectively. Although interest in vaccine development and manufacture continues to increase because of the rapid growth of the global vaccine market, this area of the bioprocess industry remains challenging and complex. Here we review the current constraints and complexities in the vaccine industry, specifically related to product development and manufacture. We…
eBook: Vaccines – Industry Collaborations to Increase Uptake
Vaccines save millions of lives every year, and the continuing increases in the number of administered doses and worldwide distribution of vaccines for long-standing diseases such as polio and malaria have contributed to the improvement in public health. Vaccine developers and manufacturers are partnering with private and government agencies to raise global vaccine uptake by addressing remaining challenges with production capacity, distribution, safety, and accessibility. And the implementation of new technologies such as virus-like particles and cell/DNA-based vaccines are helping…
Intensification of Influenza Virus Purification: From Clarified Harvest to Formulated Product in a Single Shift
Influenza is a global respiratory disease with an estimated mortality of up to a half million people per year (1). The majority of traditional influenza vaccines are still produced in eggs. Downstream processing typically consists of clarification by centrifugation, concentration by ultrafiltration, and purification by ultracentrifugation (2). Recombinant vaccines are most often purified by chromatography. Chromatographic purification of viruses already has achieved major improvements in recovery and scalability (3), but it also is important because it enables virus purification to…
Sticking In or Standing Out? Dichotomy in Vaccine Purification By Chromatography
A general vaccine purification strategy can be divided into three stages, with one or more steps for each stage. The first stage is to concentrate and isolate the target molecule quickly to remove it from conditions that could lead to its inactivation or loss. Intermediate purification seeks to remove remaining contaminants, typically using an orthogonal approach. That is followed by a polishing step in which trace impurities are removed through high-efficiency steps because those impurities usually are similar to the…
Cell-Culture–Based Influenza Vaccine Manufacturing: Evaluation of Near-Infrared Spectroscopy for In-Line Determination of Virus Titers
Each year, over 20% of the human population is infected with the influenza virus, resulting in 250,000–500,000 related deaths globally and ~38,000 deaths in the United States alone. Of further concern is the potential for pandemic outbreaks, which pose a severe worldwide threat to public health (1, 2). Vaccination has proven to be a critical tool for controlling the spread of infectious diseases, as evidenced by the eradication of polio, smallpox, and diphtheria in most parts of the world. Influenza…
Accelerated Development, Manufacturing and Monitoring of Viral Vectors
The goals of process intensification are to enhance production while shortening timelines, lessening contamination and environmental risks to products and operators, and reducing operating footprints. Previous publications from Sartorius Stedim Biotech (SSB) have highlighted key elements of such activities. In this report, the authors extend the scope of this discussion to tools and technologies that enable intensification of viral vector manufacturing processes. The first article summarizes presentations from a 2018 seminar for viral vaccine manufacturers. Three guest presentations highlighted the…
A Platform Production Process for Manufacturing Viral Vector and Vaccine Therapeutics Using Vero Cells
Yan Zhi, program design technical lead, Fujifilm Diosynth Biotechnologies Fujifilm Diosynth Biotechnologies (FDB) is a world-leading contract development and manufacturing organization (CDMO) with over 25 years of experience using biologicals and more than 310 projects including six licenses. Production sites are located in North Carolina and Texas. In Texas, the flexible biomanufacturing facility can handle production from preclinical to current good manufacturing practices (CGMP) commercial manufacturing. Facilities and capabilities include state-of-the-art process development; mobile cleanroom (MCR) technology with fully segregated…
Accelerating Vaccine Production Using a Nonviral Enabling Technology for Cell Engineering
At the recent World Vaccine Conference, Victor Ayala, PhD, an early stage investigator with Advanced BioScience Laboratories, Inc. (ABL), discussed how to accelerate vaccine production using a nonviral enabling technology for cell engineering. ABL is a contract research/manufacturing organization (CRO/CMO) providing manufacturing and laboratory research services to advance leading vaccines and therapies from clinical development to the commercial market. The company has been conducting R&D research for over 55 years and performing CMO manufacturing for over 25 years. With more…