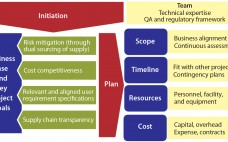
Ensuring a continuous supply of safe medicines is a key objective for the pharmaceutical industry and health authorities alike. A critical component to that end is maintaining a reliable supply of qualified raw materials (RMs) used in drug production. However, changes in suppliers, their processes, their providers, and consequently the materials they supply can occur (for a number of reasons) at any time during the life cycle of drug production. A product-supply organization therefore must be prepared to address such…
Manufacturing
Outsourcing Stability Testing: Discussions with Contract Laboratories
Stability testing is required for all biopharmaceutical drug products to detect all changes in identity, purity, and potency as a result of a number of environmental and processing factors. Whether testing is conducted in-house or through contact laboratories, it involves the development and performance of comprehensive and specific stability protocols for all stages of a product’s life cycle (1). Testing product stability in-house requires signficant time and resources, and carries challenges associated with commercialization market, time, and capacity. Market: The…
Next-Generation Bioprocessing for Meeting Healthcare Challenges: The Role Single-Use Handling Systems Can Play
The rapid spread of contagious and lethal diseases worldwide has driven bioprocess suppliers to develop technologies for use in producing disease treatments and vaccines. Bioprocessors need to develop new biologics as well as rapid and reliable methods for bringing those treatments to commercialization. Implementing modular process solutions and single‑use handling systems in closed‑manufacturing processing is one approach to addressing those needs. Developing and discovering solutions for meeting global healthcare conditions is an evolving part of bioindustry. As points of reference,…
Special Report on Assays, Test Methods, and Comparability The CMC Strategy Forum Series, Part 4, Biosimilar Products: Scientific Principles, Challenges, and Opportunities
The Chemistry, Manufacturing, and Controls (CMC) Strategy Forum held on 22 January 2012 in San Francisco, CA, focused on selected scientific and regulatory aspects in the development of biosimilar products. Such products are an increasingly important area of interest for both the biopharmaceutical industry and its regulatory agencies. Biosimilars are highly complex, so scientists have been unable to demonstrate identity to a level typically possible for small molecules. Consequently, specific scientific and regulatory approaches are required to ensure the high…
Special Report on Assays, Test Methods, and Comparability The CMC Strategy Forum Series, Part 4, The Roles of Bioactivity Assays in Lot Release and Stability Testing
A January 2007 CMC Strategy Forum on the roles of bioactivity assays in lot release and stability testing was held in Washington, DC (1). Its purpose was to promote an understanding of the design and utility of bioassays throughout product development and to delineate the conditions under which surrogate assays could be used to determine product potency. Topics of discussion included appropriate assay selection at each stage of product development, the potential use of binding assays for potency testing, and…
Quantitative Risk Assessment of Bioaccumulation Attributable to Extractables and Leachables in Cellular Immunotherapy Biomanufacturing
Precious patient samples, contamination concerns, and limited product purification options have compelled manufacturers of cellular immunotherapies (iTx) such as chimeric antigen receptor T cells (CAR-T) and T-cell receptor (TCR) technologies toward the disposables industry. Such companies are implementing single-use technologies (SUTs) almost exclusively (1). But despite the dominance of disposable bioprocess platforms and their extraordinary growth in the iTx marketplace, researchers have made limited efforts to understand the perennial and critical bioprocessing risks of leachables and extractables. Here we outline…
Effective Management of Contract Organizations: Keeping the Product Pipeline Moving, Compliant, and Available
Both small and large biopharmaceutical companies are increasingly pursuing the outsourcing of manufacturing and testing throughout the product lifecycle. The growing use of contract manufacturing organizations (CMOs) and contract testing organizations (CTOs) has led to increasing complexity within the biopharmaceutical industry as more third-party sites are leveraged to support global markets. To address those issues, a CASSS Chemistry, Manufacturing, and Controls (CMC) Strategy Forum was held in Washington, DC, 27–28 July 2014. The title was “Effective Management of Contract Organizations: Sponsors, Contract Organizations, Health Authorities and Patients — Keeping the Product Pipeline Moving, Compliant, and Available.” The CMC Strategy Forum is…
Management, Notification, and Documentation of Single-Use Systems Change Orders: Challenges and Opportunities
Single-use systems (SUS) consist of numerous plastic components derived from different suppliers. As such, they are prone to changes, including alterations in construction materials or modifications in manufacturing processes. Such changes may originate at the immediate supplier or farther back in the supply chain as a result of product improvements, process improvements, part discontinuation, or even business decisions such as manufacturing site relocation. Whether those changes are major or minor, managing their impact on biopharmaceutial processes and product quality often…
Compatibility Assessment of a Model Monoclonal Antibody Formulation in Glass and Blow–Fill–Seal Plastic Vials
Blow–fill–seal (BFS) technology has been recognized by the industry as an advanced aseptic solution (1–3). Catalent Pharma Solutions has been commercially supplying sterile BFS products to the pharmaceutical industry for decades, primarily in the respiratory and topical ophthalmic markets. Such product formulations range from simple solutions to emulsions with drug substances from classical small molecules to large complex proteins such as biologics. The company also has optimized BFS processes and its Advasept plastic container system for the manufacture of sterile…
Manufacturing Strategies for Regenerative Medicine Success
Just a few years ago, my requests for manuscripts detailing logistical considerations for cell-therapy manufacturing were met with puzzlement. The assumption seemed to be that such processes already existed for commercial biopharmaceuticals and would simply be adapted later. In quite a few cases, adopting a commercial mind-set did not appear to be a goal at all, with hands-on practitioners used to applying procedures in hospital settings. For those of us who remember early discussions about commercialization and cost-containment for protein…