Technology transfer in pharmaceutical manufacturing most often is mentioned as occurring between two organizations and involving discrete product lifecycle stages: e.g., a sponsor and a contract manufacturer and from development to manufacturing. According to the International Council on Harmonization of Technical Requirements for Pharmaceuticals for Human Use: “The goal of technology transfer activities is to transfer product and process knowledge between development and manufacturing, and within or between manufacturing sites to achieve product realization. This knowledge forms the basis for…
Manufacturing Contract Services
Specialized Outsourcing Services: Antibody–Drug Conjugates
Managing just the conjugation part of an antibody–drug conjugate (ADC) project can be a challenge. So when you consider all the component parts of an ADC, it is understandable why so many related activities are outsourced. At Piramal Grangemouth, we provide conjugation development and manufacturing services. Together with our sister sites in the United States and United Kingdom, we offer payload manufacture and fill–finish capability. The Grangemouth (UK) site has been involved in ADC conjugation services now for over 13…
Outsourcing of Buffer Preparation Activity Is Increasing
The major fluid products used in bioprocessing — culture media and buffers — are classically prepared in-house by rehydrating (dissolving and mixing) powders purchased from suppliers. Most bioprocessing facilities consider in-house preparation of these fluids to be a core bioprocessing task. However, some companies are outsourcing the work either by purchasing preprepared materials from vendors or hiring contract manufacturing organizations (CMOs) to prepare them. Buffer fluid preparation is one area of downstream production operations that are seeing an increase in…
Emerging Technology Trends in Biologics Development: A Contract Development and Manufacturing Perspective
For a contract development and manufacturing organization (CDMO), process development and manufacturing of recombinant proteins must be linked because of tight timelines driven by client expectations. Those are in turn driven by a need for rapid progression to clinical testing. Early in process development, the choice of raw materials needs to reflect existing supply chain and manufacturing infrastructure, but remain suitable for scaling up to meet future needs. One approach is to establish platform processes for a class of molecules…
Biopharmaceutical Contract Manufacturing Technology and Capacity Investments
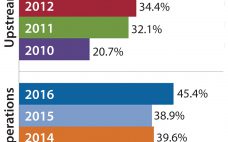
The market for biopharmaceutical contract manufacturing has shown robust growth over the past few years. The continuing growth of biopharmaceuticals and the increase in new and novel drug projects entering clinical pipelines are fueling the market’s double-digit growth rate. Responding to these new demands, contract manufacturing organizations (CMOs) are expanding both their capabilities and capacities. Background Information presented here draws from recent interviews with six executives at biopharmaceutical contract manufacturing organizations, and from HighTech Business Decisions’ latest report, Biopharmaceutical Contract…
The Virtual Pharmaceutical Company: A New Pathway to Market?
In our modern biopharmaceutical industry, the maxim “bigger is always better” no longer applies. The industry’s modest virtual model persists in its popularity, emerging as a contemporary method of workplace efficiency. With an absence of manufacturing, virtual biopharmaceutical companies have found themselves unburdened by the multiple layers of bureaucracy that often plague large companies. With such freedom, they have operated much more efficiently in terms of time, resources, product specialization, and finances. As a result, virtual biopharmaceutical companies are growing…
Outsourcing and Biomanufacturing Challenges for Emerging Therapies: A Roundtable Discussion at BIO 2016’s BPI Theater
The biopharmaceutical industry is increasingly interested in a range of emerging therapies. “We’re really starting to get beyond the monoclonal antibody,” said Patricia Seymour (senior consultant with BioProcess Technology Consultants) in her introduction to a lunchtime BPI Theater roundtable at the 2016 Biotechnology Industry Organization annual convention in San Francisco, CA, this past June. The discussion brought together three industry insiders for strategic outsourcing to talk about emerging biotherapies and their manufacturing challenges: Mark Angelino (senior vice president of pharmaceutical…
Managing Customer and Regulatory Expectations
Partnering with a contract development and manufacturing organization (CDMO) allows drug-product sponsors to turn fixed costs into variable costs. Market forecasting by pharmaceutical companies drives numerous decisions in development programs: sales-force resources, geographic resource distribution, and (of course) manufacturing planning. It is a widely accepted fact in the pharmaceutical industry that accurate forecasting is a challenge, especially for new drug launches. A number of models can be used to develop drug forecasts, but none of these models is perfect. No…
Outsourcing Trends in Biosimilars Development: A Discussion with Niall Dinwoodie (Charles River Laboratories)
No discussion about the future of the biopharmaceutical industry would be complete without assessing the impact of biosimilars. But such discussions no longer focus on whether biosimilars will enter the market, but rather when and how much market share will they take. The rapid progression of biosimilar candidates in company pipelines and the strong biosimilars research conducted by international organizations are strong indications that if your company is not already working within the biosimilars market, it may already be too…
Making the Correct Outsourcing Decisions: BPI Theater Panelists at BIO 2016 Discuss Current and Future Needs for Contract Services
Outsourcing has become a critical part of the industry in general and will play a larger role as companies continue to seek faster and more cost-effective routes to market. On Wednesday 8 June 2016, Gil Roth (president of the Pharma and Biopharma Outsourcing Association, PBOA) chaired a lunchtime roundtable titled “Making the Correct Outsourcing Decisions.” Roth’s association is a nonprofit trade group that represents CMOs and CDMOs through work with Congress and the FDA, with a focus on intra-industry issues.…