Propelled by single-use systems (SUSs), biopharmaceutical companies are approaching the ideal of continuous bioprocessing. In addition to improving process integrity and decreasing production costs, SUSs have enabled exciting ways to configure, operate, and evaluate manufacturing steps. Sensitive process analytical technologies (PATs) and discriminating data analysis platforms are supplementing those developments, helping process engineers and operators to study and modify workflows in unprecedented ways. The goal now is to intensify: to apply increasingly nuanced process knowledge and growing technological capability in…
Manufacturing Contract Services
Streamlining the Path to Clinical Manufacturing
Careful consideration of lot size is crucial for multiyear success of a cell therapy business. RoosterBio’s product design and acceleration business works with the company’s customers to help make plans that are appropriate for their stages of product and clinical development. We use the following major considerations in creating a sound multiyear strategy with intermediate milestones: Understand what the future looks like and work backward Use reasonable to conservative assumptions to estimate a range of needs Invest in the right…
CMC Development Platforms and Outsourcing to Reduce Timelines
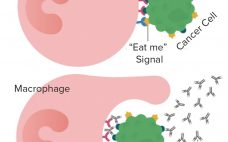
Forty Seven is a company developing novel therapies based on anti-CD47 and other immuno-oncologies. CD47 is called the “don’t eat me” signal that cancer cells give out to escape elimination by the body’s immune system. Qinghai Zhao, vice president of technical development and manufacturing, is one of the scientists working on the company’s magrolimab (5F9) monoclonal antibody that is designed to block the binding of the CD47 signal to the cell receptor SIRP-α while boosting the “eat me” signal that…
Biopharmaceutical Growth in China: Bioprocessing Challenges Are Creating CMO Opportunities
In the past decade, the world has seen rapid growth of more than 15% in China’s biopharmaceutical market (1). According to the second edition of BioPlan’s Advances in Biopharmaceutical Technology in China, the most populous country in the world — with the largest patient groups — has a growing economy with its GDP second only to that of the United States. Rapid urbanization and greater access to national healthcare insurance puts China in second place after the United States for…
Worldwide Biopharmaceutical CMO Capacity Analysis
Contract manufacturing organizations (CMOs) are a core part of the biopharmaceutical industry, with commercial manufacturing making up about a third of marketed products. Here we examine worldwide CMO biopharmaceutical manufacturing (bioprocessing) capacity, concentrating on “mainstream” CMOs — defined as those providing primarily mammalian cell culture or microbial fermentation services for manufacture at any scale of proteins and antibodies. This analysis follows our recent article examining worldwide bioprocessing capacity status and trends (1). Survey Methodology The 2018 15th Annual Report and…
A Multistep Research Protocol to Develop and Implement Validated Guidelines for CMO RFI and RFP Processes: Biopharmaceutical Vendor Evaluation and Selection Minimum Standards (BioVesel)
Pursuant to the proposal for validated minimum standards for biopharmaceutical contract manufacturing organization (CMO) request-for-information (RFI) and request-for-proposal (RFP) processes (biopharmaceutical vendor evaluation and selection minimum standards, BioVesel) (1), we propose herein a multistep research protocol to develop and implement the BioVesel standards. This proposal is intended as a basis for discussion among mulitple stakeholders. Detailed research protocols for each proposed stage in the development and implementation of BioVesel will be drafted and published separately. The context of the proposed…
Proposing a Systematic QbD Approach Toward Validated Guidelines for CMO RFI and RFP Processes: Biopharmaceutical Vendor Evaluation and Selection Minimum Standards (BioVesel)
Three major concerns predominate biotechnology executive management in organizations of all sizes and above all other risks: finance (or its absence at critical moments), technological performance, and failures in coordination. Some business functions, such as human resources (HR), are effectively siloed horizontally and therein are more likely to be susceptible to only one of those risks (1). Few functions are subject to this trinity of risks simultaneously; all functions may be exposed to failures in internal coordination, and a smaller…
Worldwide Biopharmaceutical Manufacturing Capacity Analysis: Growth Continues Across the Board
While the growth in biopharmaceutical manufacturing capacity in developed, major market countries is continuing its slow and steady climb, developing regions often are seeing double that growth rate. Over the past eight years, as detailed in the “About the Data” box, our company’s index of the top 1,000 biomanufacturing facilities (1) has tracked and ranked bioprocessing facilities worldwide in terms of known or estimated bioprocessing capacity (cumulative onsite bioreactor volume) number of biological products manufactured at clinical scale commercial scale…
Seamless Transition from R&D to Manufacturing
Fast and cheap: These criteria are becoming ever more urgent drivers for manufacturers of biologics, faced with increased scrutiny on the costs of developing novel drugs, the lengthy timelines for delivering these drugs to patients, and the tightening competition to capitalize on new targets. The challenge for manufacturers is further heightened by the expectations to deliver on quality as well. Although development and production of molecules such as monoclonal antibodies (MAbs) have greatly benefited from the “platformization” of core technologies…
Emerging Technologies and Strategies to Accelerate Process Development and Time to Market
Gustavo Mahler, president and chief executive officer, AGC Biologics AGC Biologics is one of the largest global biologics contract development and manufacturing organizations (CDMO) specializing in clinical and commercial development of therapeutic proteins. It has developed more than 200 products, both mammalian and microbial, from preclinical to commercial on all scales. The company’s CGMP facilities are located in the United States, Europe, and Japan. AGC Biologics’s technologies and strategies to accelerate time to market include standardized cell-line and cell-bank creation…