On the night of Wednesday, 27 September 2017, the fourth annual Battle of the Biotech Bands raised US$110,000 for the bands’ selected charities. More than 800 guests enjoyed refreshments at the Royale Nightclub in Boston and watched Led Zymmelin (pictured right, representing Sanofi Genzyme) walk away with the top prize. They raised money for the National Organization for Rare Disorders (NORD). The crowd was a mix of Biotech Week Boston attendees; arts and entertainment critics; developers, contractors, architects, engineers, furniture…
December 2017
December Spotlight
Revealed: Where Scientists Want to Work When you’re in the business of life-science innovation, highly trained employees are worth every penny. That’s why biotechnology companies pay top dollar for amenities-rich locations and facilities in the top US life-science real estate markets, according to Jones Lang LaSalle’s (JLL’s) Life Sciences Outlook published this past summer. JLL is a professional services firm that specializes in real estate and investment management. Its report says that life-science professionals have high expectations for their workplaces.…
Serialization: Background, Justification, Requirements, Timelines, and Readiness Across the Supply Chain
Drug manufacturers are facing unprecedented serialization challenges. Serialization requires weighty consideration and focused strategy for successful commercialization, even for those companies that have yet to bring a product to market. The World Health Organization estimates that 10% of medicines worldwide and up to 50% of drugs consumed in developing nations are counterfeit. In response to increasing drug integrity concerns, more than 40 countries have introduced laws mandating serialization and tracing of pharmaceutical products as they pass through the supply chain.…
Implementation of the BPOG Extractables Testing Protocols: Working with Multiple Single-Use Components
Single-use technologies offer significant advantages over traditional stainless-steel solutions for biopharmaceutical manufacturing. Reductions in setup times, cleaning and cleaning-validation costs, elimination of cross-contamination risks, and smaller footprints are just some of the benefits they provide. Although adoption of single-use systems (SUS) for commercial manufacturing is expanding, concerns persist that extractable and leachable (E&L) compounds from plastic SUS components potentially can leach into final drug products and compromise efficacy and safety. Those concerns are magnified amid the growing number of SUS…
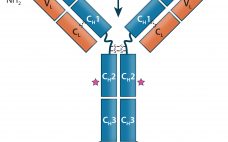
Therapeutic IgG-Like Bispecific Antibodies: Modular Versatility and Manufacturing Challenges, Part 1
Antibody-based immunotherapy has advanced significantly since 1986, when the US Food and Drug Administration (FDA) approved the first mouse monoclonal antibody (MAb) for clinical use: Orthoclone OKT-3 (muromonab-CD3). In the intervening years, researchers have applied the tools of genetic engineering to clone immunoglobulin G (IgG) genes into a number of expression vectors. In the 1990s, the bioprocess industry was able to produce fully human antibodies in cultured cells. As of June 2017, the FDA and the European Medicines Agency (EMA)…
Streamlined Column-Packing Design for a New Commercial Launch Facility
To meet network demand for a commercial launch facility, Genentech (Roche) designed a new downstream train and built it within an existing building shell at the company’s Oceanside, CA, site. This downstream train included new technologies to allow for rapid technology transfer of different new products in the company’s drug pipeline. One technology that was pursued was the Axichrom column platform from GE Healthcare and associated column packing equipment to streamline column packing design. Here we focus on how a…
Addressing the Challenge of Complex Buffer Management: An In-Line Conditioning Collaboration
Read this article from scientists at Cytiva (GE Healthcare at the time of publication) to learn more about buffer preparation strategies now. Preparation and storage of buffers is a challenge for biopharmaceutical companies developing protein-based pharmaceuticals. The need for volumes of buffer to purify increasing upstream titers have become a major bottleneck in biopharmaceutical downstream processing. Italian biopharmaceutical company Kedrion Biopharma collects and fractionates blood plasma to produce plasma-derived therapeutic products for treating and preventing serious diseases, disorders, and conditions…
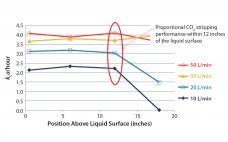
Simplify Upstream Process Development and Scale-Up: Single-Use 5:1 Turndown-Ratio Bioreactor Technology
Single-use technologies (SUTs) have been adopted widely in the biopharmaceutical industry for product development as well as clinical- and commercial-scale manufacturing. Over the years, suppliers of such equipment have addressed concerns about waste management, extractables and leachables, and reliability of supply — and as a result, end users have gained confidence in SUTs. Recognizing potential benefits that can be realized for both clinical and commercial operations, biomanufacturers increasingly are implementing SU solutions at larger scales in both upstream production and…
Drug Development in China: Lessons Learned from Years of Clinical and Small-Molecule Experience
“Today we recognize that, to successfully protect US public health, we must think, act, and engage globally. Our interests must be broader than simply those within our own borders.” —Margaret Hamburg, US Food and Drug Administration (FDA) commissioner, 2009–2015 Many major pharmaceutical companies — including Pfizer, Novartis, Eli Lilly, Sanofi, and Bayer — have research and development centers as well as clinical and commercial activities in China. Their sales revenues are increasing with the rapid growth of China’s healthcare market,…
Ask the Expert: Evaluation of the “Scale-Out” Biomanufacturing Strategy — from Early Clinical Stage to Commercialization
In a BPI Ask the Expert webinar on 2 November 2017, Jie Chen (vice president of CMC management at WuXi Biologics) spoke about scale-out biomanufacturing strategies. Chen’s Presentation Scale-out strategies allow for the use of disposable bioreactors in commercial manufacturing. A scale-out strategy offers unique advantages over traditional scale-up manufacturing: Reduces risks to product quality and process performance as manufacturing scale increases Allows for flexible process design and validation strategies Accommodates a wide range of productivity levels and market demands…