A friend of mine was returning home from a vacation in Rome this week, and her flight from Europe to Newark was delayed and diverted because someone’s child had measles on the intended connecting plane. Measles! Someone thought it was okay to spread that around. Often as we put together a themed issue — whether focused on a manufacturing pillar or something new like this month — a subtheme seems to just happen. This month, thanks to authors from Biogen…
April 2018
April Spotlight
Looking Back, Looking Ahead Check both ways before proceeding — always good advice. Industry analysts at GlobalData and Mergermarket (an Acuris company) have reviewed 2017 and made predictions for the biopharmaceutical industry in 2018. Set against a backdrop of remarkable advances in technology and the growing relevance of biosimilars, last year was marked by both disappointments and noteworthy innovations. GlobalData points to major regulatory approvals in “nearly every major therapeutic area.” These include CAR T-cell oncology treatments, the first biologic…
Cost Analysis of Cell Therapy Manufacture: Autologous Cell Therapies, Part 2
In part 2, we continue to analyze manufacturing costs of an autologous cell therapy. A typical process involves the expansion and activation of cells derived from a single patient, which is currently very labor-intensive. To date, there is little published information on overall production costs (1). In part 1, we used a software modeling platform to identify opportunities for potential cost savings. We developed a baseline model of a cell therapy manufacturing process using the production of autologous dendritic cells…
Data Science, Modeling, and Advanced PAT Tools Enable Continuous Culture
Bioprocesses traditionally use (fed-)batch cell culture processes for production of recombinant proteins and therapeutics. In batch bioprocessing, material flow is discrete, with a hold step between two unit operations, and product is harvested only once for each unit operation. Batch processes have been studied extensively and optimized through numerous advancements in experimental design (1, 2), monitoring (3–5), measurement techniques (6–9), and control strategies (10–12). However, such processes require large facility footprints for equipment (13) as well as sterilization, load, and…
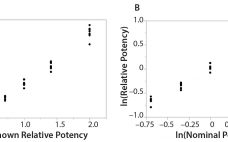
The Relationship Between R2 and Precision in Bioassay Validation
Analytical linearity along with assessments of precision and accuracy determine the range for bioassays (1). Practitioners can include coefficient of determination (R2) criteria from a linearity study in the bioassay validation protocol. Herein I illustrate the relationship of R2 to study design and analytical method variation. Overview of the Simple Linear Regression Model Dilutional linearity assesses the “ability (within a given range) of a bioassay to obtain measured relative potencies that are directly proportional to the true relative potency of the…
Scale-Up of Twin-Column Periodic Countercurrent Chromatography for MAb Purification
Periodic countercurrent (PCC) processes increasingly are being evaluated as alternatives to single-column batch capture processes. Some of the advantages of PCC processes over single-column processes include shortening of processing time and/or reduction of required resin volume through increased productivity; reduction in resin costs through improved resin capacity use; and reduction in buffer consumption through increased column loading. Those advantages, however, come with increased equipment complexity and hardware costs. PCC processes and systems with two to up 16 columns of the…
Continuous Solids-Discharging Centrifugation: A Solution to the Challenges of Clarifying High–Cell-Density Mammalian Cell Cultures
Clarification is typically the first unit operation in the purification of monoclonal antibodies (MAbs) and other proteins from mammalian cell cultures. This process removes cells and cellular debris from the culture fluid to produce a clarified cell culture supernatant that will be suitable for further purification (1). Before 2000, depth and tangential-flow microfiltration were standard clarification technologies in the biopharmaceutical industry. Process-scale centrifugation was considered to be a significant capital investment, and bioprocessors had limited ability to control (minimizing) the…
Filter-Based Clarification of Viral Vaccines and Vectors
Viral vaccines rely on the antigen properties of a virus or virus-like entity to trigger an immune response and induce immune protection against a forthcoming viral infection. Through development of recombinant viral vaccines, developers can reduce risks associated with the presence of live and inactivated viruses. Instead, recombinant vaccines induce immunity against a pathogen by relying on the capacity of one or more antigens delivered by means of viral vectors or the baculovirus/plasmid system (1). Viral vaccines are formulated with…
Ensuring the Integrity of Single-Use Containers: Providing Robustness, Science, and Helium-Based Technology with a Detection Limit of 2 μm
Identifying the greatest defect size, both for liquid leaks and microbial ingress, is a fundamental step toward protecting the integrity of single-use systems (SUS) under real process conditions. Integrity testing of such systems may become a prerequisite in the future because they are used in the most critical process steps, with detection limits correlating to liquid leaks and microbial ingress. Such testing guarantees the sterility of drug substances and drug products packaged in single-use systems and, therefore, enhance patient safety.…
Your Brand Is the Patient’s Experience
The future success of biopharmaceutical businesses will depend at least partly on their ability to create meaningful brand experiences from the start of a drug program. By “brand,” I don’t mean logos and taglines. I’m talking about meaningfully unique experiences that directly affect clinical and patient needs — specifically, to address the growing demand for self-administered injectable therapeutics. Whether you are a biosimilar developer trying to carve out differentiated value or a market leader looking at your patent protection in…