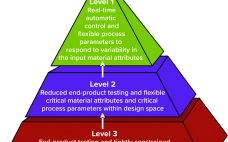
Process control enables biomanufacturers to ensure that operating parameters are within defined specifications. A control strategy should be established during early stages of process development while process and product performance are being defined using risk-based methods such as quality by design (QbD) and process analytical technologies (PATs). Confirming process control as an essential part of product development creates greater process knowledge and understanding and provides the first steps toward process optimization. By understanding how process performance relates to product quality,…
PAT
eBook: Sensor Technologies — Essential Tools for Bioprocessing 4.0
Sensors are essential devices that can be used for most, if not all, typical biopharmaceutical development and manufacturing processes to monitor fundamental process parameters such as flow, temperature, pH, and dissolved oxygen throughout all process stages. As the bioindustry progresses toward automation, digitalization, and other “Manufacturing 4.0” concepts, robust single-use and smart sensors for bioprocess monitoring will be needed. Read this BPI eBook to garner valuable perspectives on both of these types of sensors. Discussions herein focus on smart sensor…
Bioprocess Development and Qualification: PAT-Based Stage 1 and 2 Acceleration Strategies
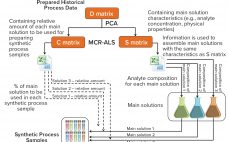
Well-established process analytical technology (PAT) strategies, such as those based on spectroscopy, bring with them several challenges related to the nature of those tools themselves (1–3). Such tools are multiparametric by design — in the sense that most spectroscopies capture multiple attributes sometimes different in nature (e.g., near-infrared, NIR, captures chemical and physical attributes simultaneously). Often a reference method is required; at other times, indirect calibrations are based on the correlation of one culture attribute with another for which a…
Trends in Data Analytics As Organizations Undergo a Digital Transformation
The biopharmaceutical industry is in the midst of an exciting transformation as biologics experience massive growth — even outpacing the small-molecule segment (1). Biologics are predicted to comprise over a quarter of the pharmaceutical market in 2020 (2). At the same time, a plethora of new biologically derived therapy concepts — e.g., cell and gene therapies — are in development. Some biologics classes have become mainstream — e.g., monoclonal antibodies — with biosimilars entering the market and contract manufacturing organizations…
eBook: Speed to IND — Balancing Risk and Reward
With so many biopharmaceuticals obtaining breakthrough or fast-track designations, companies that use accelerated strategies to be first in human studies can be left with significant quality and manufacturing challenges that must be solved later on. Despite regulatory encouragement to create solid design spaces and define parameters according to quality by design (QbD), those may go by the wayside given the pressures of speed. The reward is the investigational new drug (IND) application itself — but if companies lock in subpar…
Flow Monitoring in Continuous Processing and Single-Use Systems
Flow sensors placed at critical points in both upstream and downstream processes fulfill the regulatory goals of the process analytical technology (PAT) framework. PAT has been defined as a mechanism for design, analysis, and control of biotechnical and pharmaceutical manufacturing processes through measurement of critical process parameters (CPP). Constant flow monitoring can support its overall targets fundamentally to reduce production cycling time prevent rejection of batches enable real-time release increase automation and control improve energy and material use facilitate continuous…
Smart Sensors and Data Management Solutions for Modern Facilities
Bioprocess manufacturers continue to seek technologies for increasing productivity and shortening timelines from discovery to commercialization. Innovations such as high-throughput systems, automated platforms, and the latest clarification systems all have made processes efficient and robust. And with the increasing adoption of quality by design (QbD) principles, including the use of process analytical technologies (PAT), biomanufacturers are mitigating the risks of errors in their operations better than ever before. A critical part of mitigating risk is gathering meaningful process data and…
Control of Protein A Column Loading During Continuous Antibody Production: A Technology Overview of Real-Time Titer Measurement Methods
During production of therapeutic antibodies, harvest titer is measured to monitor product mass loaded onto the protein A capture column. This prevents both column underloading (underusing expensive resin) and overloading (wasting product as flow-through (FT)) while allowing for column yield calculations. Batch production yields a single homogenous harvest pool, thus only one titer measurement (along with volume loaded) is sufficient to determine the mass loaded. During continuous production, however, cell-free harvest (permeate) continuously exits a perfusion reactor and loads a…
Emerging Tools for Exosome Purification and In-Process Monitoring
This eBook introduces new analytical approaches that enable in-line chromatographic detection of exosomes. One approach can discriminate extracellular vesicles from nonvesicle contaminants, and one potentially can discriminate exosomes from other vesicles. Examples illustrate how they enable development of more effective and better documented purification methods. The special qualifications of monolithic chromatography media for exosome purification are discussed. New process tools designed to accommodate some of the special challenges of exosome purification are introduced. Exosomes represent one of several species of…
Multitiered Automation for Improved Efficiency of Bioprocess Analytics
The first biopharmaceutical, human insulin, was approved for use in 1982 (1). The biopharmaceutical market continues to exhibit healthy growth now, with the number of yearly patent applications increasing by 25% annually since 1995 (2). The total pharmaceutical R&D pipeline has more than doubled since the beginning of the century (Figure 1), much of that attributable to the biologics industry segment. As this industry has matured, new platform methods have emerged, and competition has increased. Consequently, the pressures of speed,…