It’s hard to believe that just six years ago, BioProcess International published its first cell therapy supplement, which included just one article on “cell therapy bioprocessing” (1). At the time, most such processing was conducted in special clinical laboratories and academic institutions. As BPI continued to cover this relatively new segment of the biopharmaceutical industry, we heard more about “the product is the process” and “scale out instead of scaling up.” After many trials, errors, and milestones, regenerative medicine has…
Analytical
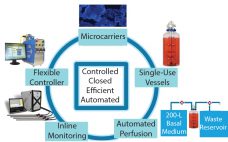
Platform Solutions for Cell Therapy Manufacturing
Advances in cell therapy have resulted in significant progress toward treating some widespread and difficult diseases, many of which represent unmet medical needs. For example, phase 3 clinical trials are already under way for therapies based on mesenchymal stem cells (MSCs), including therapies for graft-versus-host disease, acute myocardial ischemia, and chronic obstructive pulmonary disease (COPD) (1–3). Successful cell therapy treatments for such afflictions will be not only significant medical breakthroughs, but also in very high demand. However, their commercialization is…
Buffers in Biologics Manufacturing
Biotechnology has enabled commercialization of protein-based drugs including insulin, growth factors, blood factors, and antibodies. Production and purification of such biologic products require different buffers for pH control and stabilization of reactions in different steps during biomanufacture. These processes include cell culture production (the “upstream” phase), purification (the “downstream” phase), and a final phase in which excipients are introduced to the drug substance to create a drug product (“formulation and storage”). In upstream processes, buffers are primarily used for their…
Residual Host-Cell DNA in Biopharmaceutical Products: 96-Well Plate-Based Extraction and Real-Time PCR Assay for Quantitative Measurement
Regulatory guidelines that cover the development of biopharmaceutical products require testing of host-cell deoxyribonucleic acid (DNA) impurities. Real-time polymerase chain reaction (PCR) has become a popular technology for DNA quantitation and monitoring of process impurities associated with biomanufacturing. One critical challenge associated with host-cell DNA impurity testing is that recombinant proteins (e.g., monoclonal antibodies, MAbs) and their corresponding buffer components often interfere with DNA quantitation in real-time PCR reactions (1, 2). Some sample types do not require a full extraction…
A Statistical Approach to Assess and Justify Potential Product Specifications
As stated in ICH Q6B, specifications are critical quality standards that are both proposed and justified by drug product manufacturers. Xiaoyu et al. provide information on several statistically based strategies to establish specification acceptance criteria (SAC) (1). Here we address an alternative approach to relate proposed SAC for quantitative data to relevant lot history. In particular, proposed SAC can be derived in part by using calculated limits for which the lower bound of an approximate 95% confidence interval for the…
Examining Single-Use Harvest Clarification Options: A Case Study Comparing Depth-Filter Turbidities and Recoveries
Steadily increasing demand for biopharmaceutical drugs has led the industry to examine its manufacturing scales while pressuring research and development groups to produce high-yielding clones and processes. Improved media, feed supplements, bioreactor designs, and control of process parameters have helped biomanufacturers achieve multifold increases in volumetric productivity from production bioreactors. However, cell culture processes are significantly affected by their bioreactor’s ability to support cells at higher densities and sustain cultures at lower viabilities. With the implementation of a number of…
CMC Forum: Evolution of Biopharmaceutical Control Strategy Through Continued Process Verification
As defined in the ICH Q10 guideline, a control strategy is “a planned set of controls, derived from current product and process understanding, that assures process performance and product quality” (1). Every biopharmaceutical manufacturing process has an associated control strategy. FDA’s 2011 guidance for process validation (2) describes process validation activities in three stages (Figure 1). A primary goal of stage 1 is to establish a strategy for process control that ensures a commercial process consistently produces acceptable quality products.…
Response to the Publication of USP ‹1207›
The BioPhorum Operation Group’s (BPOG’s) Container Closure Integrity Testing (CCIT) workstream would like to congratulate the United States Pharmacopeia’s committee for its latest revision to USP chapter <1207> Package Integrity Evaluation: Sterile Products. Generally, we believe it provides a comprehensive overview of the available methods for container–closure testing and outlines many important elements for consideration in establishing a successful CCIT strategy. We first responded to the USP <1207> draft when it was released for comment in 2014. And from our…
HCP Antigens and Antibodies from Different CHO Cell Lines
Cell lines derived from Chinese hamster ovary (CHO) cells are widely used in therapeutic protein production because they can perform human-compatible posttranslational modifications, they are easy to use for manufacturing, and they do not propagate most human pathogenic viruses (1, 2). Expressed therapeutic proteins are secreted into CHO culture supernatant along with impurities originating from the host cells themselves. Such host cell proteins (HCPs) are important contaminants for monitoring because they directly affect drug quality, safety, and efficacy. HCPs are…
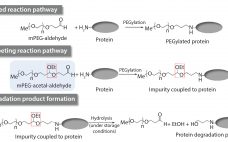
Setting Raw-Material Specifications Using Prediction Models: Determination of a Specification Limit for a Raw-Material Impurity in mPEG-Aldehyde
Impurities related to raw materials used for bioproduction can be inadvertently introduced into a manufacturing process, causing potential failure to meet in-process controls or release specifications. Unexpected impurities also can reduce yield and affect the quality, safety, and effectiveness of a final product (1). Raw-material impurities can originate from starting components or reagents used in manufacture. They can be generated in situ during synthesis or as degradation products. Impurities also can result from improper handling, packaging, and storage. Identification and…