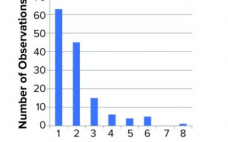
Biopharmaceuticals produced from mammalian cell cultures are susceptible to viral contamination. That risk is mitigated by applying complementary approaches. Those include extensive testing of cell banks, selecting low-risk raw materials, testing cultivations for viruses, and documenting the capacity of a purification process to inactivate and remove viral contaminants. The latter commonly is referred to as viral clearance and usually expressed as a log reduction value (LRV). Novo Nordisk has performed several viral clearance studies for different processes and process steps.…
2021
SARS-CoV-2 Hyper-Immunoglobulin: Purification and Characterization from Human Convalescent Plasma
The novel severe acute respiratory syndrome coronavirus 2 (SARS- CoV-2) emerged as a major pandemic coronavirus disease in 2019 (COVID-19) and since then has killed many people and paralyzed the global economy (1, 2). With specific antiviral therapeutic agents or antibodies yet to be approved, other antivirals and novel vaccine strategies have been essential to containing the virus and disease transmission. Passive antibody therapy can be used to limit the scope of epidemics by providing patients with antibodies that recognize…
Viral Clearance in a Downstream AAV Process: Case Study Using a Model Virus Panel and a Noninfectious Surrogate
Over the past decade, adenoassociated virus (AAV) vectors have become established as leading gene-delivery vehicles. In 2017, the pipeline for gene therapies included 351 drugs in clinical trials and 316 in preclinical development (1–4). As those candidates advance, significant efforts are being made in process development and manufacturing for viral vectors, with the overall goal of reducing process impurities while maintaining the highest possible process yield. To address that goal, industry suppliers have developed innovative AAV-specific separation technologies. Thermo Fisher…
Rapid Development of Viral Vector Production Processes: Iterative Parameter Optimization
With recent developments and successes in cell and gene therapy, the biopharmaceutical industry is facing increased demand for safe and efficient delivery systems (1). Viral vectors, including adenoviruses (AV), adenoassociated viruses (AAV), and lentiviruses (LV), are among the most common delivery agents because they infect mammalian cells efficiently. Suspension cultures have become a popular choice for robust and scalable viral manufacturing systems. Using stable cell lines that integrate all or part of the viral production elements adds further benefits by…
Ask the Expert: High-Yield mRNA Processing — From Plasmid to Highly Purified Product
Interest in industrial-scale production of messenger RNA (mRNA) has surged amid rapid development of mRNA-based vaccines against SARS-CoV-2. During an 18 February 2021 Ask the Expert presentation, Aleš Štrancar (chief executive officer of BIA Separations, a Sartorius company) reminded attendees that no platform approach yet exists for mRNA production and that much remains to be learned about manufacturing such products at commercial scales. He described current production challenges and shared BIA’s efforts to devise flexible mRNA purification tools. Štrancar’s Presentation…
Ask the Expert: Leveraging Cloud Capabilities to Execute CHO Cell Culture Experiments
Process development teams have relied for decades on benchtop bioreactors to perform mammalian-cell–culture experiments; however, doing so requires significant resources and staffing. During a 24 February 2021 presentation, Deborah Pascoe, PhD, vice president of operations at Culture Biosciences (Culture), explained how to leverage her company’s high-throughput, cloud-connected bioreactors to execute Chinese hamster ovary (CHO) cell cultures with high reproducibility and scalability. Pascoe’s Presentation Pascoe described her company as an extension of a customer’s laboratory. After technology transfer, Culture and a…
Defining a Successful Biotechnology Entrepreneur
Having brought its first vaccine candidates from concept to clinical development in less than three months, BioNTech is making global waves as the pioneer behind Pfizer’s COVID-19 vaccine. Because of such success stories, more eyes than ever are looking at opportunities raised by the biotechnology industry. As talented people around the world from academia, clinical practice, and other industries consider careers in this sector, they should examine the characteristics necessary to become successful biotechnology entrepreneurs. Biotechnology fuses science and business.…
eBook: Buffers — Navigating New Demands on Downstream Raw Materials
Bioreactor titers for monoclonal antibody (MAb) processes have increased significantly since the dawn of the biopharmaceutical industry, yet such gains have instigated bottlenecks for critical high-volume raw materials used in downstream processing, such as buffer solutions. As downstream purification is required for most, if not all, biopharmaceutical products, buffers and their preparation are topics that concern nearly every drug company. But those topics rarely receive direct attention. This BPI eBook explores what factors prompted the current buffer bottleneck and what…
How to Improve the Capturing of Antibody Fragments
Some of the latest promising biopharmaceutical drug substances are antibody fragments. Antibody fragments are either separate functional subunits of antibodies or recombinant molecules, which, just like antibodies, are composed of immunoglobulin domains. These drugs offer several therapeutic advantages over conventional monoclonal antibodies. Upstream processing for antibody fragments is easier than it is for standard antibodies. Recombinant-based antibody fragments can be modified to meet specific needs of affinity, avidity, valence, and action mode. They also can be produced in prokaryotic cells…
Introduction: Technologies Converge in Biopharmaceutical Laboratories
Bioassay development is foundational to the well-characterized biotechnology product paradigm. Bioassays are the best tools for drug developers to use in determining the biological activity (potency) of their products, which has been a biopharmaceutical critical quality attribute (CQA) since long before that concept had a name. Thus, these assays are vital to quality assurance and quality control (QA/QC), preclinical studies, and clinical testing — and by extension to process development and monitoring. Because of their complex nature, bioassays are among…