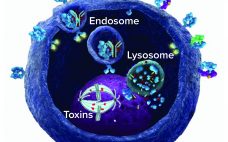
G-protein–coupled receptors (GPCRs) are a large and diverse family of seven-transmembrane–domain proteins expressed on the surface of human cells. These molecules respond to external stimuli by initiating signal-transduction pathways that affect the expression of a large family of genes — which, in turn, regulate a range of vital physiological processes and functions. Figure 1 illustrates the general pathways of GPCRs. Without these proteins, humans simply could not survive: Without β-adrenergic receptors, we could not regulate our blood sugar, for example;…
2020
October 2020: From the Editor
This issue has been created by editors living in over-the-top levels of wildfire smoke here in Oregon’s Willamette Valley. Although all of us have been safe, we are appalled at the widespread loss of property and lives over these past two weeks. Amid those losses, and against the backdrop of political and social upheavals, the coronavirus continues to wreak its own havoc on just about every aspect of our lives. Adding natural disasters and subsequent loss of homes and possessions…
The Crossroads of Academia, Industry, and Education: Modern Training Centers Are Pivotal to the Future of R&D
Global pharmaceutical industry research and development (R&D) investment has experienced steady growth over the past two decades, with an anticipated compound annual growth rate (CAGR) of 3.0% and projected 2024 investment of US$213 billion (1). Focused on developing innovative therapies for chronic, infectious, genetic, and lifestyle-related ailments, the fast-growing biologics segment has become a cornerstone of the pharmaceutical industry and healthcare sector. The demonstrated effectiveness and wide-ranging applicability of biopharmaceuticals also have brought considerable R&D in computational and biological technologies.…
Next-Generation Biotechnology Product Development, Manufacturing, and Control Strategies, Part 1: Upstream and Downstream Strategies
Future biomanufacturing must address industry drivers, including the need for decreasing cost of goods (CoG), increasing market globalization, shortening development time for pipeline products, reducing risk to patient supply, and improving product quality. A CASSS chemistry, manufacturing, and controls (CMC) forum entitled “Next-Generation Biotechnology Product Development, Manufacturing, and Control Strategies” took place on 16–17 July 2018 in Gaithersburg, MD, to address those opportunities. Advanced technologies include single-use bioreactors, alternating tangential-flow (ATF) systems used during fermentation, modular and closed process equipment,…
Development of a Single-Use Hermetic Centrifuge System for Mammalian Harvest with Moderate to High Cell Content
The production of increasingly higher cell densities has stressed the already limited solids-handling capabilities for traditional intermittent ejection centrifuge systems. By contrast, a single-use disc-stack centrifuge based on the solids-flow principle offers distinct advantages for cell culture harvesting. Such benefits include solids handling of high-density cell culture processes and elimination of the separation disruption and aerosol generation associated with the intermittent solids ejection. A single-use system also provides well-established benefits of disposable components — such as removal of steam- and…
A New Runway for Purification of Messenger RNA
A high-performing capture method is a critical bedrock asset for developing industrial purification processes. This is especially true for extended families of products that share highly similar chemical composition. Therapeutic monoclonal IgG is an example. The ability of protein A affinity chromatography to achieve 95% purity in one simple step was the runway that got recombinant immunotherapy off the ground and made it available to millions. In fact, protein A did more. Beyond giving the industry a foundation manufacturing method,…
How Much Harm Can a Single Droplet Do? Considerations for a Viral Inactivation Step
Viral clearance is a fundamental aspect of viral safety for biopharmaceutical products. Regulatory agencies around the world require biomanufacturers to segregate their operations appropriately to mitigate the risks of carryover contamination from previous process steps or product batches and of crossover contamination between product(s) made in the same facility. Guidelines are vague in defining “appropriate,” leaving biomanufacturers to interpret regulatory expectations and define their own virus reduction and segregation strategies. Given the differences among manufacturing processes and facilities housing such…
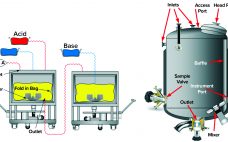
Dye-Stripping Buffer and Resin Stripped-Dye Analysis: Development and Optimization of a Novel Spectrophotometric Assay and Method for Removal of Cibacron Blue Dye
Biopharmaceutical process-related impurities encompass all organic and inorganic materials that arise from the biomanufacturing process apart from the drug substance. According to ICH Q6B guidelines on test procedures and acceptance criteria for biotechnological products, these impurities include cell substrates such as host-cell proteins, host-cell DNA, endotoxins, and so on; inducers, antibiotics, media components, and chromatographic media used in purification processes; and solvents and buffer components (1). Impurities can cause undesired, deleterious pharmacological effects if ingested or injected by a patient,…
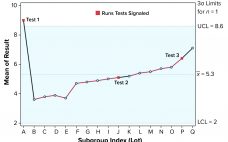
Run Rules with Autocorrelated Data for Continued Process Verification
Control charts can be used to assist in monitoring of biopharmaceutical product quality attributes as part of continued process verification activities. A number of tests known as run rules have been developed to assess whether biomanufacturing processes remain in statistical control. In practice, results for such attributes can be positively autocorrelated. Simulated data are used to assess the performance of run rules with autocorrelated data to assist in determining risk–reward profiles for process monitoring. Autocorrelated Data The tendency for data…
Quality Risk Management for Filter Integrity Testing: Compliance with EMA’s Future Annex 1
Quality risk management (QRM) is a systematic process for assessment, control, communication, and review of risks to the quality of a pharmaceutical product across its lifecycle. Although QRM is not new (1), the regulatory focus on QRM will increase with the arrival of the European Medicines Agency’s (EMA’s) Annex 1 (2), which was reviewed by the US Food and Drug Administration (FDA), the World Health Organization (WHO), and the Pharmaceutical Inspection Convention Scheme (PIC/S). Integrity testing of sterilizing-grade filters is…