My first business trip each year usually is to Washington, DC, in January. There, the CMC Strategy Forum and WCBP symposium (sponsored by CASSS have an excellent track record of setting the stage for topics that will play out through the rest the year. The combined event draws significant US Food and Drug Administration participation. So we also look forward to their help in focusing our attention on key regulatory issues and updates. With continuing focus on well-characterized biologics, WCBP…
March 2020
New Directions in Bioprocess Development and Manufacturing
The biopharmaceutical industry continues to grow at a rapid pace. That growth is accelerating even more because of keen investor and research interests in cell and gene therapies. My focus below is on several key growth areas in the field, specifically aspects of development and biomanufacturing. This discussion builds on my 2018 column on top trends in bioprocessing (1). In that article, I identified five key trends that continue to resonate now: rapid development of high-productivity upstream processes advances in…
Scalable Manufacturing of Lentiviral Gene Therapies
Lysosomal storage diseases are caused by mutated genes that express defective lysosomal proteins, such as essential enzymes. For example, cystinosis is a metabolic disease caused by a defective gene that encodes cystinosin, an exporter protein (Figure 1). Avrobio develops gene therapies to treat lysosomal storage diseases. On 8 October 2019, the company announced that its first patient had been dosed in the AVR-RD-04 investigational gene therapy program, which involves genetic modification of the patient’s own hematopoietic stem cells to treat cystinosis.…
The Next Global Life-Science Leader
Canada’s life-science industry is exceptional at producing innovative research. With less than 0.5% of the world’s population, our country produces 5% of the globe’s total research publications. Canada’s citation rate ranks among the top six nations and is 43% higher than the global average (1). At the same time, our life-science industry is competing with other global jurisdictions into which investment is flowing. The US state of Massachusetts, already the number one biotechnology cluster in the world, has committed US$1.5…
Using Blockchain Technology to Ensure Data Integrity: Applying Hyperledger Fabric to Biomanufacturing
Digitalization of manufacturing operations is a major challenge that many industries face. With the advent of smart equipment, automation of unit operations and complete processes, and digitalization of batch documentation, more data are generated now than ever before. The information must remain manageable, and data integrity needs to be ensured. The challenge for biomanufacturers will be to ensure that their entire large output of data will be attributable, legible, contemporaneous, original, and accurate (ALCOA) as defined by the US Food…
A Response Plan for Viral Contamination in Bioproduction Facilities
The biopharmaceutical industry uses living biological systems as a platform for manufacturing of protein-based drugs, vaccines, and other therapies derived from or consisting of different cell types. On one hand, living systems are inherently susceptible to viral infection and may harbor endogenous viruses, so the potential for such contamination cannot be eliminated. On the other hand, the industry has an excellent patient-safety record. Viral safety is achieved through three fundamental measures: prevention (e.g., by selection), removal (by clearance and/or reduction),…
Implementation of Single-Use Miniature Bioreactors to Support Intensified Cell Culture: Using Functional Performance Indicators to Assess a Small‑Scale Model
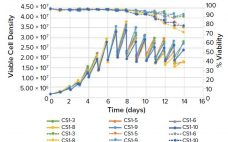
Changes to bioprocessing in the biopharmaceutical industry are driven by the need for increased speed, lower cost of goods (CoG), and greater flexibility (1). To meet these challenges, the industry is adopting strategies that include intensified processing. During the initial stages of intensified processing, it is essential to identify the most productive and/or stable clones for use before starting pilot-scale studies. That requires screening large numbers of clones and then further testing the most promising ones in benchtop bioreactors. The…
Ask the Expert: Accelerating Development and Manufacturing Platforms for Viral Vectors
Bai-wei Gu, Juan Lagos, and Matthew Weaver (heads of cell line development, upstream process development, and downstream process development groups, respectively, at WuXi Advanced Therapies, ATU) joined forces on 29 October 2019 to feature their company’s viral-vector manufacturing capabilities for cell and gene therapies. In addition to adherent platforms for lentivirus (LV) and adenoassociated virus (AAV) vectors, ATU soon will offer suspension-cultured viral vector platforms for them as well as analytical measures that support release testing. Transitioning from adherent to…
Ask the Expert: Best Practices for Aseptic Sampling from Stainless-Steel Equipment
Turn-key single-use aseptic sampling devices (ASDs) have diminished bioprocess contamination risks significantly. But depending on testing, facility, and storage needs, some ASD container types are more effective than others are. Bobbi Allen (technology expert at Sartorius Stedim Biotech North America, SSB) focused her 8 January 2020 “Ask the Expert” presentation on “what, why, when, and where” operators must sample aseptically from stainless-steel tanks. Using data from in-house testing of aseptic sampling containers, Allen offered key considerations for sterility, process monitoring,…
Better Bioprinting Ahead: Breakthroughs and Remaining Challenges
Bioprinted organs soon could revolutionize clinical trials, transplantation, and regenerative medicine. But as Chris Lo reminds us in a new GlobalData report (1), several technical hurdles must be negotiated before biopharmaceutical companies can harness three-dimensional (3D) bioprinting for such purposes. BPI explores persistent printing problems and promising solutions below by analyzing Lo’s report alongside commentary from founding editorial advisory board member Bill Whitford (bioprocess strategic solutions leader at GE Healthcare Life Sciences), Lev Gerlovin (vice president in the life sciences…