I am writing this shortly after BPI’s publisher Brian Caine and I conducted some interviews at an east-coast supplier company. Our experience there reinforced our rationale — from the start of this publication — for continually seeking ways to build on the collegial, collaborative nature of relationships in our industry. I’ve been covering this industry as an editor since 1989; Brian, Cheryl Scott, and Maribel Rios have been involved since the mid-1990s — so we have seen technologies come and…
April 2015
Spotlight
Niche-Disease: Pompe’s Disease by Cheryl Scott Pompe’s disease is a genetic disorder that causes a cellular buildup of the complex sugar glycogen. Its accumulation in organs and tissues impairs their ability to function normally. Since the disorder was identified in 1932 by Dutch pathologist J.C. Pompe, researchers have classified three forms of Pompe disease (also called glycogen storage disease type II or acid maltase deficiency): classic infantile-onset, nonclassic infantile-onset, and late-onset. The disease is inherited in an autosomal recessive pattern.…
Outsourced Data Integrity: Are Short-Term Financial Gains Worth Long-Term Headaches?
Competitive pricing and continued cost pressures have contributed to the need for many US biopharmaceutical companies to outsource manufacture of active pharmaceutical ingredients (API) and finished products from countries with lower costs for labor, material, and equipment. The main benefit of doing so is lower costs of manufacturing with quality standards comparable to those found in the United States. India and China now account for 80% of API production. But those countries have received media attention because of biopharmaceutical manufacturing…
When Is a Virtual Business Model Suitable for Biopharmaceutical Companies?
Virtual companies are based on the model that all activities are outsourced. Such companies have no (or few) employees, occupy no laboratory space, and use contract service organizations for all activities. Over the past decade, several virtual biopharmaceutical companies have formed (1–4). They are primarily start-up ventures that use contract research organizations (CROs) for R&D and contract manufacturing organizations (CMOs) for product manufacturing. By contrast, a fully integrated biopharmaceutical company is based on the model that all activities are internal to…
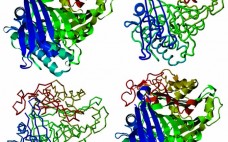
Hamster Phospholipase B-Like 2 (PLBL2): A Host-Cell Protein Impurity in Therapeutic Monoclonal Antibodies Derived from Chinese Hamster Ovary Cells
All recombinant protein biotherapeutics must be tested for the presence of residual host-cell protein (HCP) impurities (1–3). The most common analytical method for doing so is a polyclonal sandwich immunoassay. Polyclonal anti-HCP antibodies are selected to recognize the broadest population of HCPs possible. The immunogen and analytical standard are produced from a blank-run fermentation that mimics the production run but lacks the specific biotherapeutic protein. Because of the large number of impurities present in harvested cell-culture fluid (HCCF) that might…
Rapid Development and Scale-Up of Biosimilar Trastuzumab: A Case Study of Integrated Cell Line and Process Development
Compared with that for new drugs, biosimilar development faces significantly condensed timelines from cell line to first-in-human (FIH) trials. A biosimilar development program needs to accelerate quickly toward preclinical and phase 1 studies; phase 2 studies typically are not required because dose response and other patient-treatment concepts are already established by the original, comparator medicine. Phase 3 studies typically are limited to fewer patients, which ultimately shortens overall timelines and costs. The key challenge remains: demonstrating comparability and high similarity…
Bioreactor Design for Adherent Cell Culture: The Bolt-On Bioreactor Project, Part 2 — Process Automation
The Bolt-on Bioreactor (BoB) project is an independent initiative to develop and commercialize a bioreactor for automated and efficient culture of adherent cells, especially in production of therapeutic cells and other biopharmaceuticals (1). After conducting thorough research on available culture systems for adherent cells, the BoB team believes that a successful alternative to existing devices must solve four major challenges. Addressed in the first installment of this series (2), the first challenge concerns volumetric productivity. The second challenge is…
Building a Robust Biological Assay for Potency Measurement
Potency is a critical quality attribute of a biological product and is often determined by a biological assay (also called bioassay or biopotency assay). Specifically, potency is the biological activity or capacity of a product directly linked to its clinical efficacy. Potency tests are performed as part of product release, comparability studies, and stability testing. Nonbiological methods — which measure a product’s molecular or biochemical characteristics (e.g., ligand-binding assay) — have gained interest as replacements for often troublesome bioassays. Even…
Single-Use Processing for Microbial Fermentations
During the past decade, single‑use bioprocessing has emerged as a standard platform for current good manufacturing practice (CGMP) mammalian cell culture. Biomanufacturers have come to appreciate the benefits of lower capital and operating costs, reduced contamination risk, continuity from early development through manufacturing, flexibility, and sustainability (1). Disposable cell‑culture vessels have gained wide acceptance because their performance duplicates that of stainless‑steel, fixed‑tank bioreactors, with which manufacturers have extensive experience. This is no accident: Single‑use bioreactors use stainless–steel engineering principles, particularly…
Culture Media and Protein Expression From Conversations with William G. Whitford
As part of BPI’s “Ask the Expert” series, editorial advisor William G. Whitford (senior technical market manager for GE Healthcare Life Sciences) spoke with editor-in-chief Anne Montgomery and marketing and digital content strategist Leah Rosin on two separate occasions about issues related to culture media and expression titers. Sourcing Serum-Free Media Anne discussed cell culture media and process fluids with Bill in March 2014. Whitford: Things are advancing, and the industry is changing significantly. In general, we are using more…