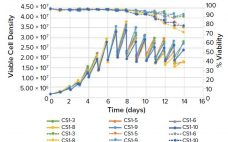
Changes to bioprocessing in the biopharmaceutical industry are driven by the need for increased speed, lower cost of goods (CoG), and greater flexibility (1). To meet these challenges, the industry is adopting strategies that include intensified processing. During the initial stages of intensified processing, it is essential to identify the most productive and/or stable clones for use before starting pilot-scale studies. That requires screening large numbers of clones and then further testing the most promising ones in benchtop bioreactors. The…
Sunday November 16, 2025