Advances in cell therapy have resulted in significant progress toward treating some widespread and difficult diseases, many of which represent unmet medical needs. For example, phase 3 clinical trials are already under way for therapies based on mesenchymal stem cells (MSCs), including therapies for graft-versus-host disease, acute myocardial ischemia, and chronic obstructive pulmonary disease (COPD) (1–3). Successful cell therapy treatments for such afflictions will be not only significant medical breakthroughs, but also in very high demand. However, their commercialization is…
Author Archives: Eytan Abraham
Meeting Lot-Size Challenges of Manufacturing Adherent Cells for Therapy
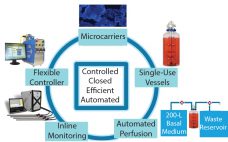
Adherent cells such as adult primary cell lines and human multipotent (MSCs) and pluripotent stem cells (hPSCs) present a manufacturing challenge as lot sizes increase from 109 (billions) to 1012 (trillions) cells (1). Typically, manufacturing platforms are good for one log of expansion. So new methods will be required to achieve commercially relevant lot sizes. Traditional two-dimensional culture methods have been used to grow anchorage-dependent cell types. Although such methods are reliable and well defined, they are very labor intensive…
Meeting Lot-Size Challenges of Manufacturing Adherent Cells for Therapy
Adherent cells such as adult primary cell lines and human multipotent (MSCs) and pluripotent stem cells (hPSCs) present a manufacturing challenge as lot sizes increase from 109 (billions) to 1012 (trillions) cells (1). Typically, manufacturing platforms are good for one log of expansion. So new methods will be required to achieve commercially relevant lot sizes. Traditional two-dimensional culture methods have been used to grow anchorage-dependent cell types. Although such methods are reliable and well defined, they are very…