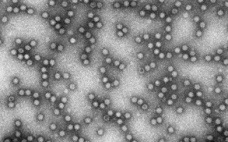
Part 1 of this two-part report describes an investigation into the potential cause(s) and ways to control a product quality attribute (PQA) of a protein expressed in perfusion cell culture (1). The presence of low–molecular-weight (LMW) species following size-exclusion high-performance liquid chromatography (SEC-HPLC) is a protein quality attribute that can indicate an increase in truncated forms of the expressed protein and/or other LMW moieties. The expressed protein in this study is a heavily glycosylated recombinant glycoprotein (rGP) comprising two subunits:…
Thursday, December 17, 2020 Daily Archives
Practical Considerations for Statistical Analyses in Continued Process Verification
Several statistical techniques can be used to assist in monitoring biopharmaceutical product quality attributes as part of continued process verification (CPV) activities. These include run charts, control charts, and capability analyses. Below, I provide an overview and recommendations on statistical strategies when developing a CPV program, considering the expected behavior of manufacturing results in the biopharmaceutical industry. Presence of Autocorrelated Data In a previous study, I highlighted the tendency for data to be positively autocorrelated (values are closely related to…
The Watershed Moment for ADCs Has Arrived — Now, CDMO Selection Is the Last Barrier
As far back as 10 years ago, antibody–drug conjugates (ADCs) were being talked about as the next big breakthrough in pharmaceuticals due to their highly targeted approach to therapies, especially in areas such as oncology. When Adcetris (brentuximab vedotin) became the second ADC approved by the US Food and Drug Administration (FDA) in 2011, it was predicted to be a watershed moment when the floodgates would open for more approvals and novel ADC approaches. Despite that possibility, the approval of…
Analytical Support for Biologics: A Conversation with Almac Sciences
Almac Sciences (a member of the Almac Group) recently expanded its suite of analytical solutions to include biologics testing. This follows a 2019 expansion of the company’s facility in Athlone, Ireland, where it provides a comprehensive range of flexible pharmaceutical testing services to support drug development programs adhering to industry regulations and good manufacturing practice (GMP) standards. “Biologics have gained huge traction in the past decade and are poised for stronger growth in the coming years with potential to significantly…
Ultrasonic Flow and Bubble Sensors: Optimize Process Quality in Single-Use Bioprocessing Applications
Process monitoring in laboratory and production environments enables continuous control and optimization of critical process parameters. Hence, the early detection of errors is an effective means of increasing process efficiency and reproducibility, improving quality and safety parameters, and reducing long-term costs. Highly precise and flexible noncontact clamp-on flow and bubble sensors are useful instruments to fulfill these goals. They can be applied effectively to buffer and media preparation, chromatography and filtration systems, bioreactors and fermentors, feed and transfer lines, and…
Evolving Business Models to Meet the Future of Healthcare
Yourway provides integrated supply-chain solutions for the global pharmaceutical and biotechnology industries. We exceed traditional offerings to provide customized support that includes warehousing and packaging, project management, planning and optimization guidance, comparator sourcing, ancillary-supply sourcing, forecasting, and returns/reconciliation management. Our single–specialized-provider approach offers clients a high level of convenience and efficiency that translates into quality, speed, and operational improvements. A Growing Global Footprint As we introduce new added-value solutions, expand our global footprint, and continue investing in infrastructure, technology, training,…
Ask the Expert: Predicting Viral Clearance During Downstream Development
Until recently, downstream process development (PD) teams have lacked methods for easy, effective, and economical estimation of a process’s viral clearance capability. David Cetlin (senior director of R&D at Cygnus Technologies) delivered an “Ask the Expert” presentation on 13 October 2020 describing how MockV kits containing noninfectious mock-virus particles (MVPs) could fill that gap. Cetlin’s Presentation Viral clearance studies tend to be outsourced to contract research organizations (CROs) because they require biosafety level (BSL)-2 and -3 facilities for working with…
rBIO launches aiming to increase manufacturing output of insulin
rBIO plans to lower cost of insulin by 30% through the ‘hyper-expression’ of newly-developed strains of bacteria. The start-up company is launched out of San Francisco and arrives alongside the news that it has successfully synthetically produced human insulin, using E. coli bacteria as the host for recombinant DNA synthesis. According to rBIO, the next stages for the biotech are to upscale production of insulin and to identify other prescription drugs that can be manufactured using its approach. Already the…
Inovio contracts Kaneka for plasmid DNA COVID vaccine
Inovio has partnered with plasmid manufacturer Kaneka Eurogentec to support the production of its COVID-19 vaccine candidate, INO-4800. Kaneka Eurogentec, an affiliate of Kaneka Corporation, will join Inovio’s growing network of contract development and manufacturing organizations (CDMOs) to support INO-4800, a COVID-19 plasmid DNA-based vaccine, set to move into a Phase II/III clinical trial. The biotech has not disclosed the terms of agreement. However, Inovio’s CEO, Joseph Kim, said that “Kaneka Eurogentec will be a crucial member of Inovio’s global manufacturing…
Some cold chain issues with Pfizer’s COVID vaccine but US confident of supplies
US military officials say several trays of Pfizer and BioNTech’s COVID-19 vaccine needed to be replaced after being stored at excessively cold temperatures. Details of the replacement were disclosed yesterday during a press conference about efforts to roll out the vaccine. General Gustave F. Perna, who is running the US’ Operation Warp Speed (OWS), said some vaccine sent to California and Alabama was replaced after it was stored at a temperature lower than the recommended 70°C. “We had two trays…